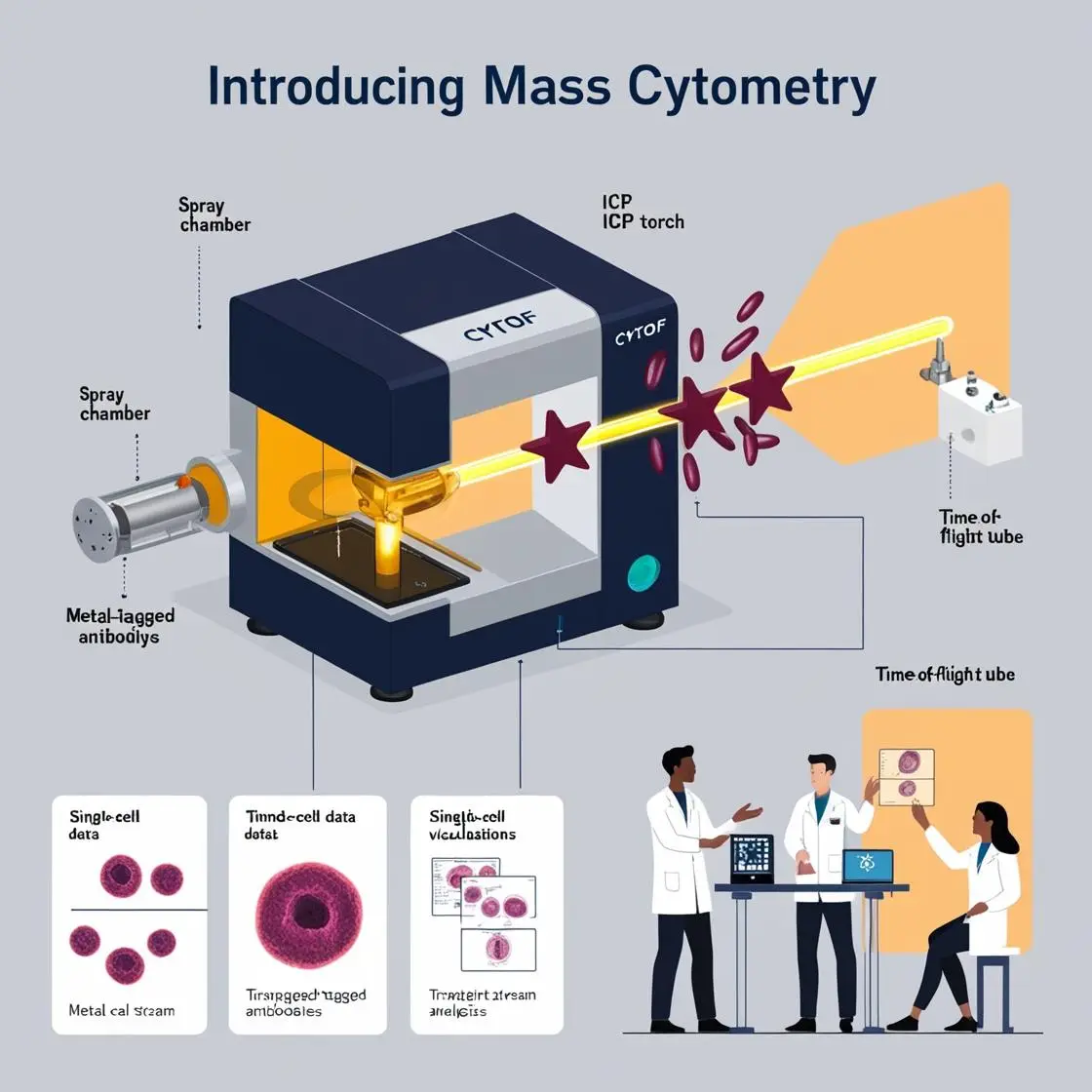
The development and evaluation of vaccines have been revolutionized by the advent of high-dimensional technologies like mass cytometry. This chapter explores how CyTOF has transformed our understanding of vaccine-induced immune responses, the identification of correlates of protection, and the optimization of vaccine formulations.
Assessing Vaccine-Induced Immune Responses
Mass cytometry has provided unprecedented insights into the complexity of vaccine-induced immune responses. The landmark study by Newell et al. (2013) in Immunity, “Cytometry by time-of-flight shows combinatorial cytokine expression and virus-specific cell niches within a continuum of CD8+ T cell phenotypes,” demonstrated the power of CyTOF in revealing the intricate landscape of T cell responses following vaccination. This work showed that vaccine-induced T cells exist in a continuum of phenotypes rather than discrete subsets, challenging our previous understanding of cellular immunity.
In the context of COVID-19 vaccine development, Arunachalam et al. (2021) published a pivotal study in Nature cited over 1100 times, “Systems biological assessment of immunity to mild versus severe COVID-19 infection in humans.” Using high-dimensional flow cytometry, they characterized immune responses to SARS-CoV-2 infection and vaccination, providing crucial insights that informed vaccine development strategies.
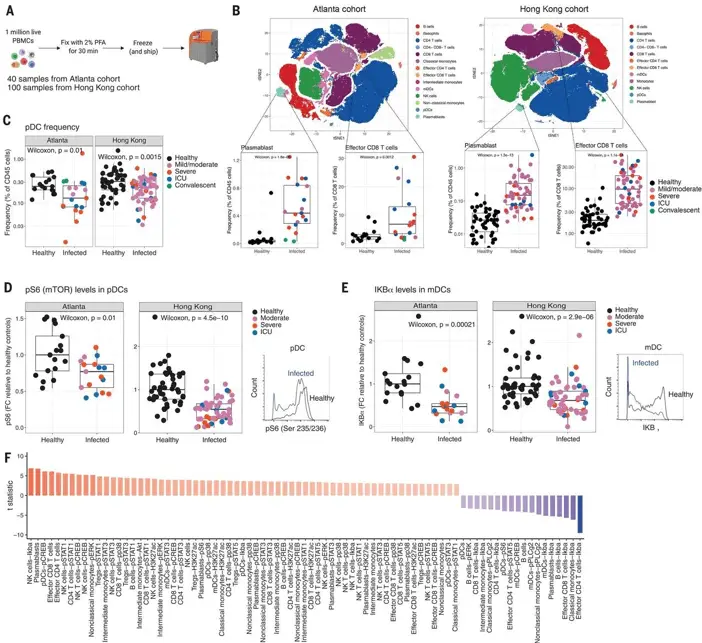
Identifying Correlates of Protection
One of the most challenging aspects of vaccine development is identifying reliable correlates of protection. CyTOF has proven invaluable in this pursuit.
A study by Kotliarov et al. (2020) in Nature Medicine, “Broad immune activation underlies shared set point signatures for vaccine responsiveness in healthy individuals and disease activity in patients with lupus,” used CITE-seq to identify shared immune signatures that predict vaccine responsiveness. This work has implications not only for vaccine development but also for understanding autoimmune diseases.
Cancer Vaccines and Epitope Discovery
In cancer vaccine development, the identification of tumor-specific epitopes is crucial. CyTOF has been instrumental in characterizing T cell responses to these epitopes.
The rapid development of COVID-19 vaccines provides an interesting parallel to cancer vaccine research. The mRNA vaccine platform, used by Moderna and Pfizer-BioNTech, allowed for the rapid design of vaccine candidates. Remarkably, Moderna designed its mRNA-1273 vaccine just 66 days after the SARS-CoV-2 genetic sequence was published (Corbett et al., 2020, Nature, cited 1500 times).
Interestingly, the development of these vaccines relied heavily on computational approaches. While not specifically using R, bioinformatics tools were crucial in identifying potential epitopes and predicting their immunogenicity. This in silico approach is increasingly being applied to cancer vaccine development, where identifying tumor-specific neoantigens is a key challenge.
CyTOF in Vaccine Development: Three Key Studies
- Palgen et al. (2018) in Nature Communications, “Prime and Boost Vaccination Elicit a Distinct Innate Myeloid Cell Response,” used CyTOF to characterize innate immune responses to vaccination, revealing how different vaccine strategies activate distinct myeloid cell populations. The analysis was performed with SPADE and SPADEVizR.
- Apostolidis et al. (2021), in Nature Medicine, wrote a research cited over 300 times “Cellular and humoral immune responses following SARS-CoV-2 mRNA vaccination in patients with multiple sclerosis on anti-CD20 therapy“. They implemented high-dimensional flow cytometry analysis of circulating T cell populations after SARS-CoV-2 mRNA vaccination, They used opt-SNE, an advanced version of tSNE. The researchers revealed that while antibody and B cell responses were reduced in patients suffering from multiple sclerosis on anti-CD20 therapy, they still generated robust antigen-specific CD4 and CD8 T cell responses. Interestingly, anti-CD20 treatment skewed the immune response, compromising follicular helper T cell responses but augmenting CD8 T cell induction. This study demonstrates how high dimensional flow cytometry can provide crucial insights into vaccine-induced immune responses in immunosuppressed populations, potentially guiding vaccination strategies for vulnerable groups.
- Since T-cell play a key role in cancer immunity, high dimensional analysis is a key tool in cancer vaccine development, as shown in the last study of Gohil et al. (2021) “Applying high-dimensional single-cell technologies to the analysis of cancer immunotherapy”.
Industrial Applications
Several pharmaceutical companies have leveraged CyTOF in their vaccine development programs:
- Moderna: While Moderna is known for its rapid development of the mRNA-1273 COVID-19 vaccine, they have also used mass cytometry in their research. In a study by Lindgren et al. (2017) published in Frontiers, “Induction of Robust B Cell Responses after Influenza mRNA Vaccination Is Accompanied by Circulating Hemagglutinin-Specific ICOS+ PD-1+ CXCR3+ T Follicular Helper Cells,” Moderna researchers used CyTOF to characterize immune responses to mRNA vaccines, demonstrating the technology’s value in their vaccine development pipeline.
- Genentech: Researchers at Genentech have utilized CyTOF in various studies. For instance, Bjornson-Hooper et al. (2019) published a paper in Cell Reports, “A comprehensive atlas of immunological differences between humans, mice and non-human primates,” which used mass cytometry to compare immune system compositions across species, providing crucial insights for translational research in vaccine development.
- GlaxoSmithKline (GSK): GSK has employed CyTOF in their research, as shown by an online video https://videos.flow.standardbio.com/watch/PFt8GttYdrTF4Lgc7SNK4u
Patel, Investigator at the CyTOF Preclinical Capability lead GSK, discusses the implementation of CyTOF technology at GSK, highlighting its impact on research, including deep immune profiling of exhausted CD8+ T cells, and how the team has expanded and streamlined its use. The introduction of user-friendly and automated CyTOF XT™ systems has positively influenced drug discovery programs across various stages.
Mass cytometry has transformed vaccine development and evaluation, providing unprecedented insights into the complexity of vaccine-induced immune responses. From characterizing T cell phenotypes to identifying correlates of protection and optimizing vaccine formulations, CyTOF has become an indispensable tool in vaccinology. As we continue to face new infectious diseases and push the boundaries of cancer immunotherapy, the high-dimensional data provided by CyTOF will undoubtedly play a crucial role in developing the next generation of vaccines. The rapid development of COVID-19 vaccines has demonstrated the power of integrating computational approaches with advanced immunological techniques, a synergy that promises to accelerate vaccine development across a range of diseases. As we look to the future, the combination of in silico prediction, high-dimensional immune profiling, and rapid vaccine platforms offers hope for quicker responses to emerging threats and more personalized approaches to cancer immunotherapy.
Who would've thought that typing code could be as crucial as pipetting cells? When I first dipped my toes into the world of R, I felt like a fish out of water. But soon, I realized it was more like being a fish discovering it could breathe on land. Coding isn't just for computer geeks anymore. It's the Swiss Army knife of modern science. Take the COVID-19 vaccine - it wasn't just cooked up in a lab, it was predicted by algorithms first. That's right, our silicon friends gave us a head start in the race against the virus. Some might argue that scientists should stick to science and leave coding to the pros. But that's like saying you don't need to write because you have a secretary. By learning to code, I've not only discovered new things, I've actually started to enjoy it.
Guillaume Beyrend