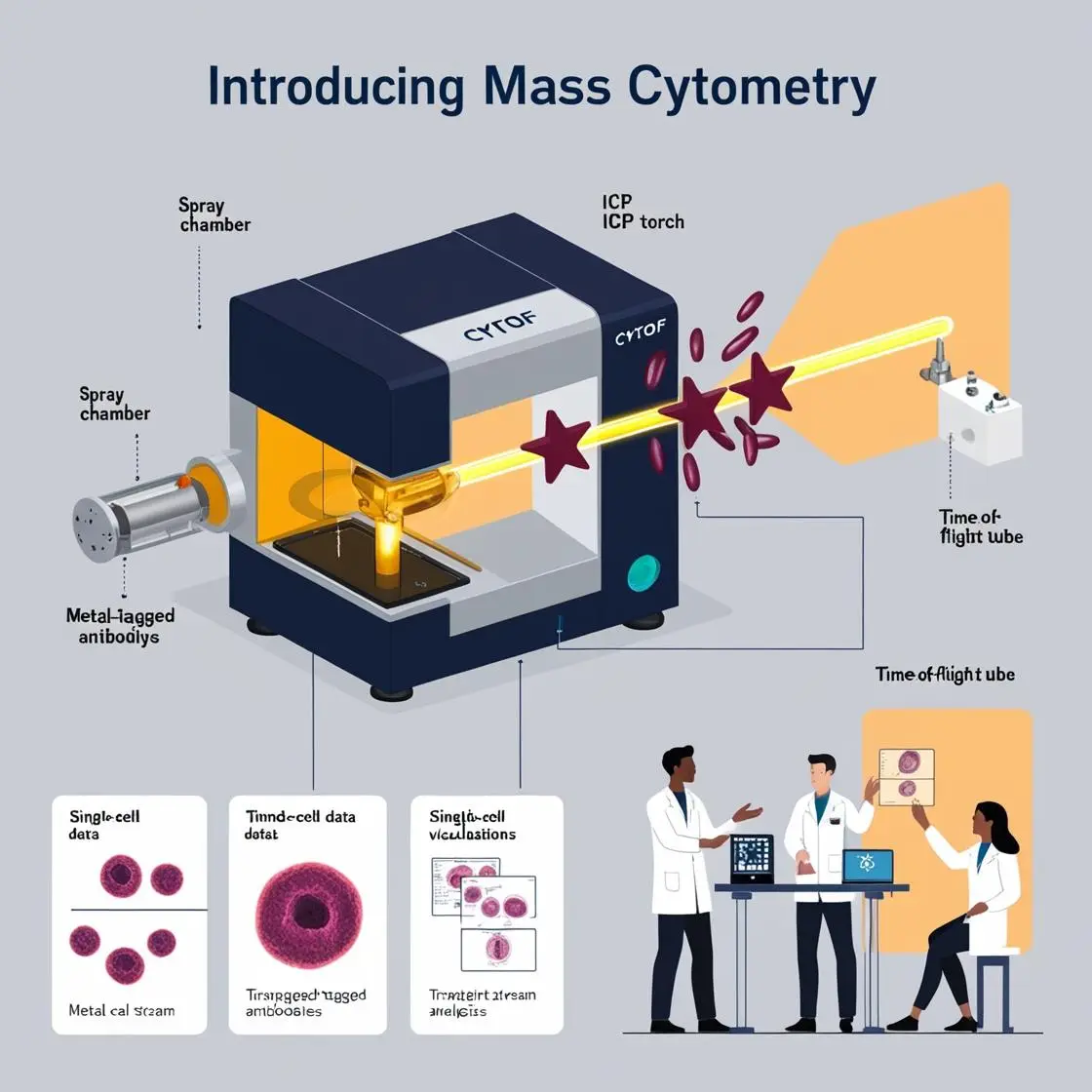
The complex ecosystem of solid tumors has long challenged our understanding of cancer biology and treatment. Mass cytometry has emerged as a powerful tool to dissect this intricate microenvironment, offering unprecedented insights into the cellular composition and interactions within tumors. Let’s explore how this technology revolutionizes our understanding of solid tumor biology.
Characterizing Immune Infiltrates
Mass cytometry has dramatically enhanced our ability to characterize tumors’ diverse immune cell populations. The high-dimensional nature of the data allows for the identification of rare and novel cell subsets that may play crucial roles in tumor progression or response to therapy.
- Chevrier et al. (2017) in Cell, “An Immune Atlas of Clear Cell Renal Cell Carcinoma,” used a merged 73-marker CyTOF panel to create a comprehensive map of the immune landscape in renal cell carcinoma. This study revealed unexpected immune cell populations and their spatial distributions, providing new insights into tumor-immune interactions.
- Lavin et al. (2017) in Cell, “Innate Immune Landscape in Early Lung Adenocarcinoma by Paired Single-Cell Analyses,” employed CyTOF to profile early-stage lung tumors, uncovering a unique immune signature that could predict patient outcomes. This work demonstrated the power of mass cytometry in identifying prognostic immune markers in early-stage cancers.
Stromal Cell Populations in Tumors
Beyond immune cells, mass cytometry has illuminated the diverse stromal cell populations within tumors, revealing their critical roles in tumor progression and therapy resistance.
- Wagner et al. (2019) in Cell, “A single-cell atlas of the tumor and immune ecosystem of human breast cancer,” used CyTOF alongside other single-cell technologies to create a comprehensive atlas of breast cancer. This study revealed novel stromal cell populations and their interactions with tumor cells, providing a new framework for understanding breast cancer heterogeneity.
- Izar et al. (2020) in Cell, “A single-cell landscape of high-grade serous ovarian cancer,” employed CyTOF to characterize both tumor and stromal cells in ovarian cancer. This work uncovered previously unrecognized stromal cell subtypes associated with patient outcomes, highlighting the importance of the tumor microenvironment in disease progression.
All of those authors were cited over a hundred times, showing such discoveries’ significant impact.
Spatial Organization of Tumor Ecosystems
The development of imaging mass cytometry has added a crucial spatial dimension to our understanding of tumor ecosystems, allowing for the visualization of cellular interactions within the tissue context.
- Giesen et al. (2014) in Nature Methods, “Highly multiplexed imaging of tumor tissues with subcellular resolution by mass cytometry,” introduced imaging mass cytometry, demonstrating its potential for high-dimensional tissue imaging. This technological breakthrough paved the way for spatial analysis of tumor microenvironments.
- Jackson et al. (2020) in Nature, “The single-cell pathology landscape of breast cancer,” used imaging mass cytometry to predict the effects of immunotherapy in cancer,
Recent Breakthroughs
In a recent study, Hoch et al. (2022) in Science, “Multiplexed imaging mass cytometry of the chemokine milieu in melanoma characterizes features of the response to immunotherapy” used mass cytometry to predict immunotherapy responses. They found that CXCL9 and CXCL10 were present in patches with CXCL13+ exhausted T cells, suggesting that they recruited B cells and aided in forming tertiary lymphoid structures (TLS) in melanoma tumors. TLS had a spatial enrichment of naïve and naïve-like T cells involved in antitumor responses.
How CyTOF Enabled These Discoveries
Mass cytometry has been instrumental in these groundbreaking studies for several reasons:
- High-Dimensional Analysis: The ability to simultaneously measure 40+ proteins at the single-cell level has allowed for identifying complex cellular phenotypes that were previously indistinguishable.
- Rare Cell Detection: CyTOF’s sensitivity enables the detection and characterization of rare cell populations that may play crucial roles in tumor biology.
- Unbiased Exploration: The high-parameter nature of CyTOF data allows for unbiased clustering approaches, leading to the discovery of novel cell subsets.
- Integrative Analysis: CyTOF data can be easily integrated with other single-cell technologies, providing a more comprehensive view of tumor ecosystems.
- Spatial Context: Imaging mass cytometry has added crucial spatial information, allowing researchers to understand cellular interactions within the tissue architecture.
The application of mass cytometry to solid tumor analysis has fundamentally transformed our understanding of cancer biology. From uncovering novel immune and stromal cell populations to revealing the spatial organization of tumor ecosystems, CyTOF has provided unprecedented insights into the complexity of cancer. These discoveries are not mere academic exercises; they have profound implications for cancer diagnosis, prognosis, and treatment. As we continue to leverage the power of mass cytometry, we move closer to unraveling the intricate tapestry of cellular interactions that define cancer, paving the way for more effective, personalized therapies. The tumor microenvironment, once an impenetrable fortress, is now yielding its secrets to the penetrating gaze of mass cytometry, offering new hope in our ongoing battle against cancer.
In the whirlwind world of tumor research, we stumbled upon a cellular superhero: the TAI cells. As we reported in our paper, "PD-L1 blockade engages tumor-infiltrating lymphocytes to co-express targetable activating and inhibitory receptors" (Beyrend et al., 2019), these T cells Activated and Inhibitory were quite the discovery. The name TAI was a stroke of bilingual brilliance. You see, I did my PhD in the Netherlands, where "taai" means "tough" or "tenacious". And these cells? They wore that name like a badge of honor. Our paper showed that these TAI cells, co-expressing both activating and inhibitory receptors, were the immune system's elite force against cancer. It was like discovering that some T cells had been secretly training for the cellular equivalent of the Olympics. So there you have it – TAI cells, the tough cookies of the immune world, proving that in science, a great discovery can also come with a great name. Who said research can't have a sense of humor?
Guillaume Beyrend