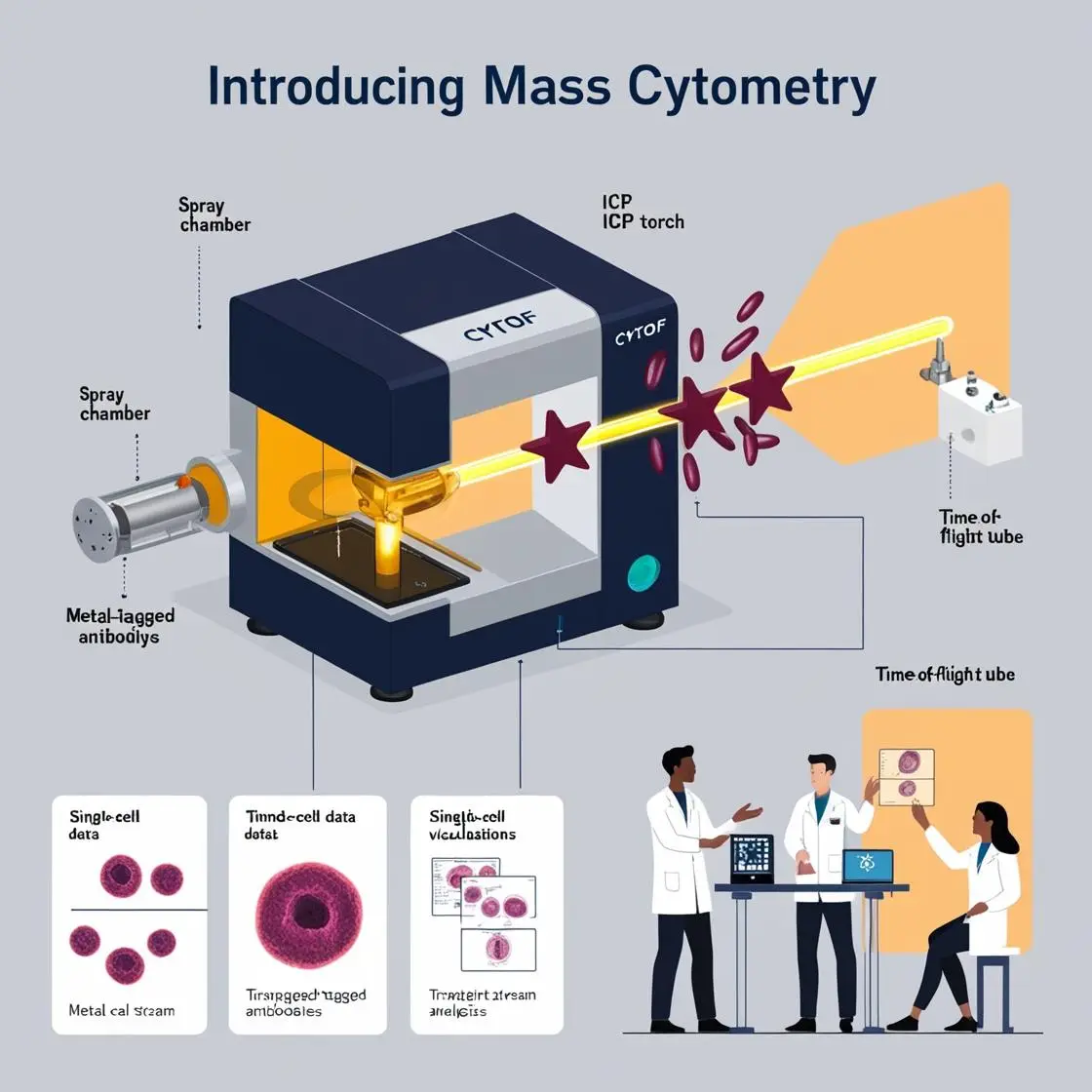
Organ transplantation has been revolutionized by the advent of mass cytometry (CyTOF), offering unprecedented insights into the complex immunological landscape of transplant recipients. This technology is reshaping our understanding of graft rejection, tolerance, and immunosuppression management.
Monitoring Graft Rejection and Tolerance
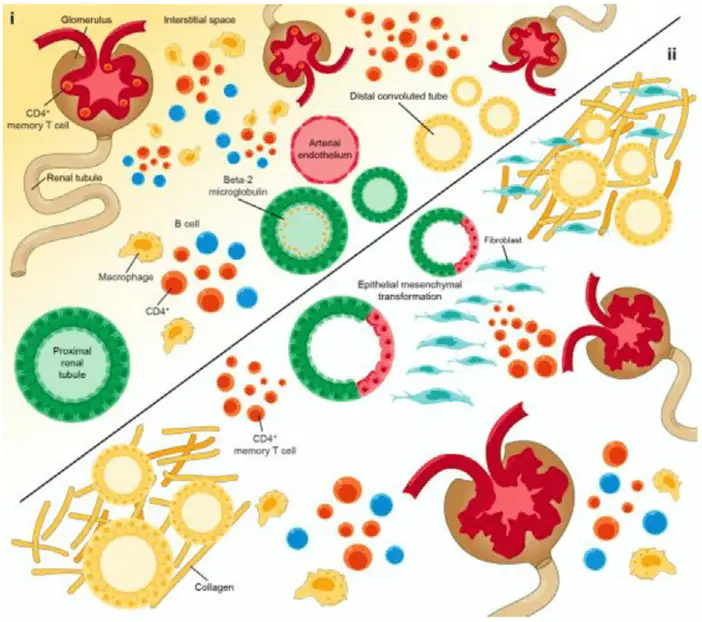
CyTOF has dramatically enhanced our ability to predict and monitor graft rejection. The very recent study by Alexander et al. (2024) in American Journal Transplantation used imaging mass cytometry (IMC) to analyze 32 kidney transplant biopsies with various types of allograft inflammation, utilizing a panel of 28 biomarkers to explore the immune landscape at the single-cell level. The researchers developed novel analytical tools for cell segmentation and classification, including a kidney tissue microstructure classifier, which allowed for detailed analysis of immune cell distribution within different kidney compartments. Using PhenoGraph clustering, they identified 11 immune and 9 nonimmune cell clusters, revealing that memory T cells and macrophages were prevalent across different types of allograft inflammation. The study also employed machine learning to identify spatial biomarkers that could discriminate between different inflammatory phenotypes. Notably, the researchers found that certain cell clusters, such as those representing epithelial-mesenchymal transformation and memory T cells, correlated with poor graft outcomes. This exploratory study demonstrates the potential of IMC to provide a more comprehensive understanding of kidney allograft inflammation, which could lead to improved diagnostic accuracy and personalized treatment strategies in transplant medicine.
Anecdote: Consider the case of Sarah Hyland, the actress known for her role in “Modern Family,” who received a kidney transplant in 2012. Had CyTOF been routinely available then, her post-transplant journey might have been different. If we had CyTOF when she first got her transplant, doctors could have seen early signs of rejection in her blood before she felt any symptoms. It’s amazing to think how this technology could have helped tailor treatments and potentially avoided some of the complications she faced.”
Optimizing Immunosuppression Regimens
CyTOF’s ability to simultaneously analyze dozens of cellular markers has transformed our understanding of donor-specific immune responses. A study by Liu et al. (2021) in Frontiers used CyTOF to analyze immune cell profiles in peripheral blood samples from five kidney transplant recipients before and after immunosuppressive treatment. The researchers identified significant changes in various immune cell populations, including decreases in central memory CD4+ T cells, follicular helper CD4+ T cells, effector CD8+ T cells, effector memory CD8+ T cells, and naive CD8+ T cells, as well as an increase in CD57+ NK cells after immunosuppressive therapy. These findings provide insights into the dynamic changes in the immune landscape following kidney transplantation and immunosuppressive treatment, potentially offering guidance for more accurate assessment and treatment strategies for kidney transplant recipients.
Anecdote: Apple co-founder Steve Jobs, who underwent a liver transplant in 2009, faced significant challenges with immunosuppression. Studying his blood with CyTOF technology might have helped him in this respect.
Real-World Impact of CyTOF in Transplantation
The impact of CyTOF on transplantation immunology is profound:
- Early Rejection Detection: CyTOF can identify subtle changes in immune cell populations that precede clinical signs of rejection, allowing for earlier intervention.
- Personalized Immunosuppression: By providing a detailed map of a patient’s immune landscape, CyTOF enables truly personalized immunosuppression regimens.
- Tolerance Monitoring: CyTOF can identify immune signatures associated with transplant tolerance, potentially allowing some patients to reduce or even discontinue immunosuppression safely.
- Novel Therapeutic Targets: The high-dimensional data provided by CyTOF is revealing new cellular subsets and signaling pathways that could be targeted for more effective anti-rejection therapies.
As CyTOF technology continues to advance and become more widely available, its impact on transplant medicine will only grow. From reducing the risk of rejection to minimizing the side effects of immunosuppression, CyTOF is helping to ensure that the gift of organ donation translates into lasting health and improved quality of life for recipients.
The future of transplantation, illuminated by the high-dimensional insights of CyTOF, is brighter than ever. As we continue to decode the intricate cellular orchestration of the immune response to transplanted organs, we move closer to a world where organ rejection becomes a rarity and transplant recipients can live life to the fullest. CyTOF is not just a tool for research; it’s a beacon of hope for transplant patients worldwide, promising a future where the harmony between donor organs and recipient immune systems is no longer a distant dream but an achievable reality.
Transplantation immunology might not have been my direct focus, but at the LUMC in the Netherlands, where I did my PhD, I was breathing the air of immunological history. I worked in the same department where Jan van Rood discovered the HLA system - talk about standing on the shoulders of giants! Leiden, it turns out, is a veritable hotbed of medical breakthroughs. Take Willem Einthoven, who invented the electrocardiogram (EKG) right here in Leiden, giving us a window into the heart's electrical soul. Or consider the discovery of Factor V Leiden, a genetic quirk in blood clotting that put this city on the hematological map. And let's not forget Anton van Leeuwenhoek, who peered through his pioneering microscopes in Leiden, opening up the world of microorganisms to human eyes for the first time. It's like Leiden has been playing a centuries-long game of "Eureka!" bingo.
Guillaume Beyrend