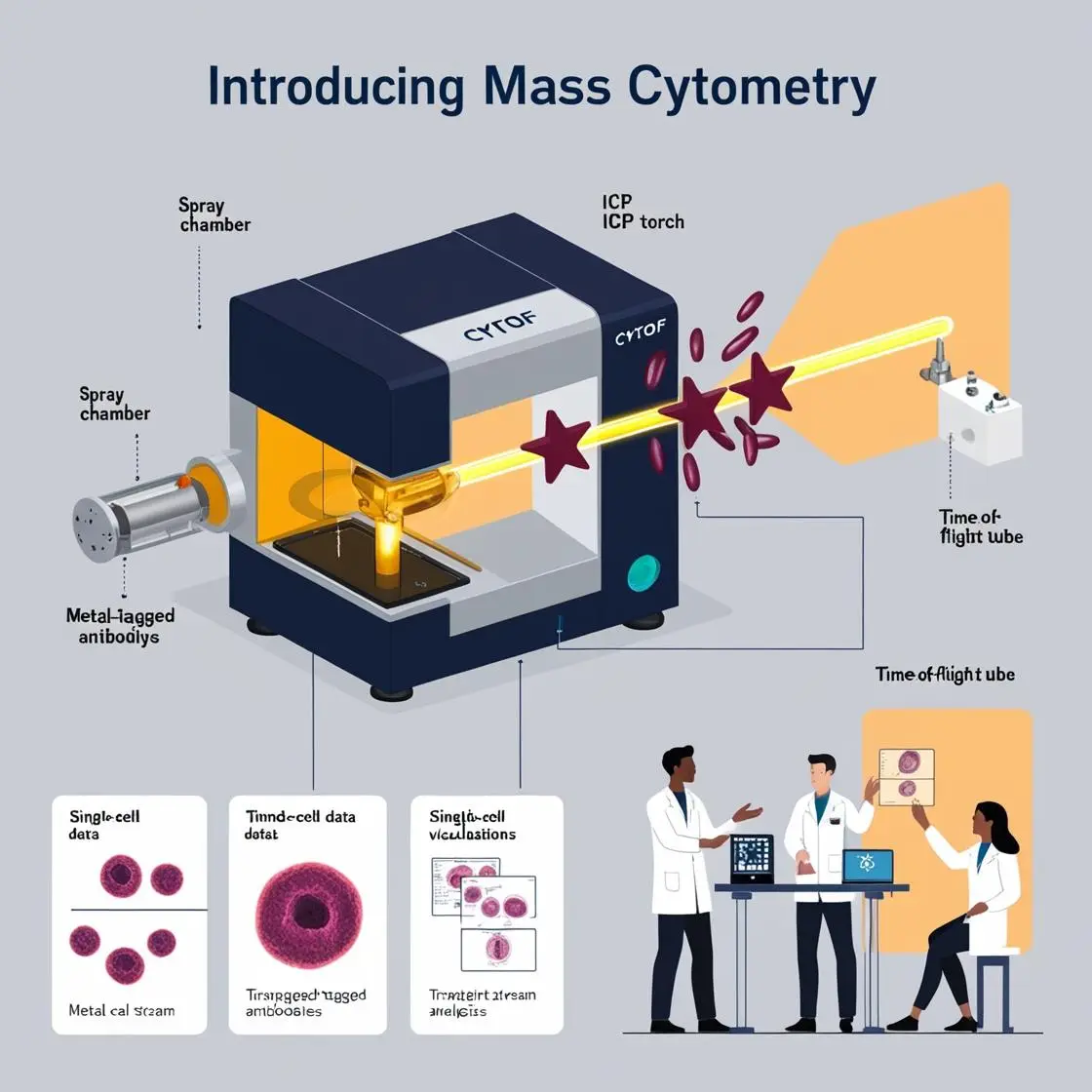
Tracking Cellular Changes During Therapy
Mass cytometry allows us to observe the evolving cellular landscape of tumors during treatment, providing crucial insights into therapeutic efficacy and potential resistance mechanisms.
A landmark study by Leelatian et al. (2017) in Cytometry Part B: Clinical Cytometry demonstrated the utility of mass cytometry for analyzing human tissues and solid tumors. This work laid the groundwork for using CyTOF to track cellular changes during therapy in clinical settings.
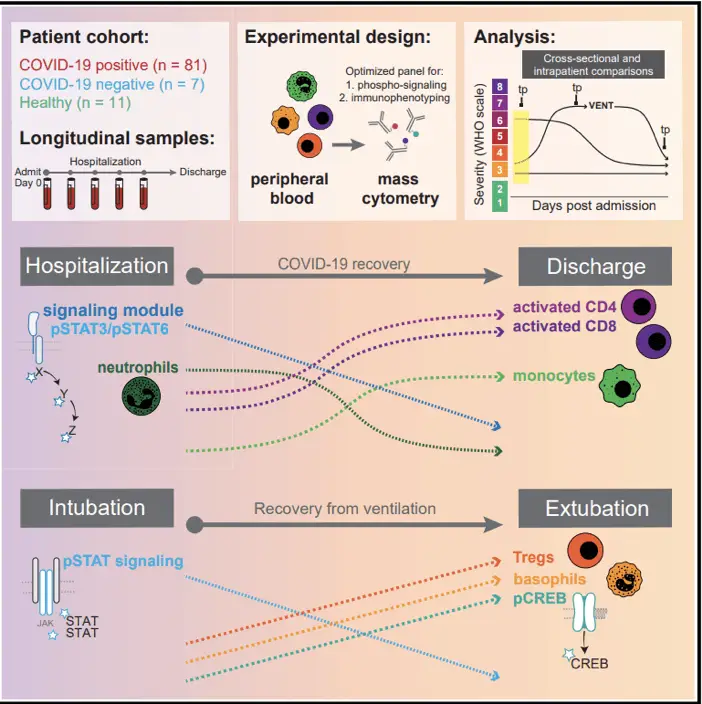
A recent study published by Burnett et al. in Immunity (2022) demonstrated that mass cytometry can identify a consistent immune recovery pathway in hospitalized COVID-19 patients. Analysis over time within individual patients revealed shifts in myeloid and T cell levels and a decrease in immune cell communication across different types of cells as the disease improved and patients were discharged. These changes, along with an increase in regulatory T cells and decreased signaling in basophils, were also seen in patients recovering from respiratory failure and were linked to better outcomes upon admission. Despite variations in patients’ immune profiles and disease progression, a common immune pathway characterizes recovery from severe SARS-CoV-2 infection.
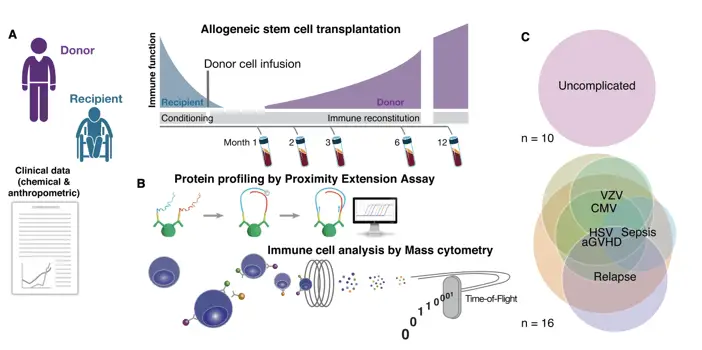
Lakshmikanth et al. (2017) in Cell demonstrated the power of mass cytometry in tracking immune changes during therapy. Their study on allogeneic stem cell transplantation combined mass cytometry with serum profiling to analyze 26 patients over 12 months. Using machine learning and topological data analysis, they uncovered global immune signatures predicting clinical outcomes, despite the small, heterogeneous patient group. This approach revealed coordinated changes across immune cells and proteins, offering insights into post-transplantation immune system regeneration. The study showcases how mass cytometry, paired with advanced analytics, can transform our ability to monitor and predict therapeutic outcomes in complex immunological scenarios, paving the way for more personalized immunotherapies.
Identification of Immunotherapy Resistance Mechanisms
Mass cytometry has played a crucial role in uncovering resistance mechanisms to immunotherapy. A landmark study by Wei et al. (2017) published in Cell, titled “Distinct Cellular Mechanisms Underlie Anti-CTLA-4 and Anti-PD-1 Checkpoint Blockade,” used mass cytometry to analyze tumor-infiltrating T cells in patients treated with checkpoint inhibitors. They identified distinct cellular mechanisms of action for anti-CTLA-4 and anti-PD-1 therapies, providing insights into why some patients respond differently to these treatments.
Further advancing our understanding, Spitzer et al. (2017) in Cell, “Systemic Immunity Is Required for Effective Cancer Immunotherapy,” employed mass cytometry to reveal how systemic immune responses contribute to immunotherapy efficacy. Their work highlighted the importance of considering the entire immune landscape, not just tumor-infiltrating cells, when studying resistance mechanisms.
A recent study by Wang et al. (April 2024), published in Nature Communications, presents a systematic approach to identifying potential chemo-immunotherapy combinations to overcome anti-PD-1 resistance in cancer. The researchers developed a “shift ability score” to measure how chemotherapy or small molecule inhibitors might alter anti-PD-1 resistance in tumor cells. They mapped the landscape of chemo-immunotherapy synergism by analyzing transcriptomics data from over 41,000 compounds and 16,000 shRNA-treated cancer cell lines. The study uncovered a mitochondrial RNA-dependent mechanism for drug-induced immune activation in tumors. This comprehensive in silico screening approach offers a new strategy for identifying and characterizing mechanisms of immunotherapy resistance, potentially accelerating the development of more effective combination therapies to overcome resistance in cancer patients.
Practical Examples in Hospital Settings
- Neoadjuvant Therapy Monitoring:
For breast cancer patients receiving neoadjuvant chemotherapy, serial biopsies analyzed by mass cytometry could track changes in tumor-infiltrating lymphocytes. A study by Gonzalez-Ericsson et al. (2021) in Clinical Cancer Research used this approach to identify immune signatures associated with pathological complete response, potentially allowing for real-time adjustment of treatment strategies.
- Minimal Residual Disease Detection:
Mass cytometry’s high sensitivity in leukemia treatment allows for the detection of minimal residual disease (MRD) at levels not possible with conventional flow cytometry.
- Immunotherapy Response Prediction:
Mass cytometry analysis of tumor biopsies for melanoma patients considering immunotherapy could help predict response. Krieg et al. (2018) in Nature used CyTOF to identify a specific myeloid cell signature in the blood that predicted response to anti-PD-1 therapy, potentially guiding treatment decisions.
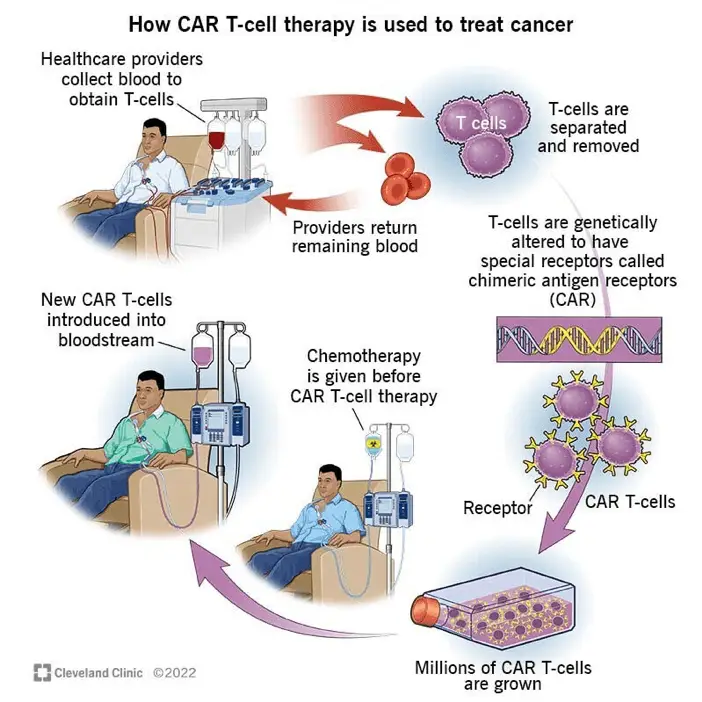
CAR-T Cell Therapy and Mass Cytometry: A Powerful Partnership
Chimeric Antigen Receptor T-cell (CAR-T) therapy represents one of the most exciting breakthroughs in cancer treatment in recent years. Mass cytometry has played a crucial role in developing, optimizing, and monitoring CAR-T cell therapies.
Understanding CAR-T Cell Heterogeneity:
Mass cytometry’s high-dimensional capabilities have been instrumental in revealing the heterogeneity of CAR-T cell products. A landmark study by Fraietta et al. (2018), cited over 1000 times, in Nature Medicine used CyTOF to identify a specific subset of CD8+ T cells associated with complete responses in chronic lymphocytic leukemia patients treated with CAR-T therapy. This level of detailed characterization would not have been possible with conventional flow cytometry.
Monitoring CAR-T Persistence and Evolution:
The ability to simultaneously measure dozens of parameters allows researchers to track the fate of CAR-T cells in patients over time. Sheih et al. (2020) in Nature Medicine used mass cytometry to monitor CAR-T cell populations in patients with B-cell malignancies. Their paper, “Clonal kinetics and single-cell transcriptional profiling of CAR-T cells in patients undergoing CD19 CAR-T immunotherapy,” employed mass cytometry alongside other techniques to track CAR-T cell differentiation and expansion. They identified distinct CAR-T cell subsets associated with response durability and demonstrated how mass cytometry can provide insights into CAR-T cell behavior in vivo. Similarly, in 2023, Louie et al. showed in Nature Communications that CAR+ and CAR- T cells share a differentiation trajectory into an NK-like subset after CD19 CAR T cell infusion in patients with B cell malignancies. These studies showcase how mass cytometry can be used to monitor and characterize CAR-T cells in patients, providing valuable information about treatment efficacy and potential mechanisms of response or resistance.
Analyzing the Tumor Microenvironment:
Mass cytometry enables comprehensive profiling of the tumor microenvironment before and after CAR-T therapy. This has been crucial in understanding how the native immune system interacts with CAR-T cells and how the tumor environment changes in response to treatment. For instance, Locke et al. (2024) in Nature Medicine used flow cytometry to reveal the “impact of tumor microenvironment on the efficacy of anti-CD 19 CAR T cell therapy or chemotherapy and transplant in large B cell lymphoma”. Based on the phase 3 ZUMA-7 trial, the research found that high B cell gene expression signatures and CD19 expression were associated with improved outcomes for axicabtagene ciloleucel (axi-cel) therapy, but not for standard of care.
Industrial Applications and Clinical Impact:
Several biotechnology companies have leveraged mass cytometry in their CAR-T cell research and development:
- Novartis: In developing Kymriah, the first FDA-approved CAR-T therapy, Novartis used mass cytometry to characterize their CAR-T product. This is evidenced in a paper by Corneau et al. (2021) published in the British Society for Haematology, which describes using mass cytometry to characterize CAR-T cells and develop a detection method.
- Kite Pharma (a Gilead company): Kite Pharma has utilized mass cytometry in their CAR-T research. This is demonstrated in a study by Good et al. (2022) published in Nature Medicine, which used mass cytometry to characterize CAR-T cell products and correlate cellular phenotypes with clinical outcomes.
Clinical Impact:
The insights gained from mass cytometry have directly impacted patient care in CAR-T therapy:
- Patient Selection: By identifying biomarkers associated with response, clinicians can better select patients likely to benefit from CAR-T therapy.
- Product Optimization: Understanding which T cell subsets are most effective has led to improvements in CAR-T manufacturing, potentially enhancing efficacy and reducing side effects.
- Toxicity Management: Mass cytometry has helped reveal mechanisms behind CAR-T-associated toxicities like cytokine release syndrome, leading to better management strategies.
- Combination Therapies: Insights into the tumor microenvironment have suggested potential combination therapies to enhance CAR-T efficacy, such as checkpoint inhibitors.
Real-world Example:
Consider a patient with relapsed/refractory diffuse large B-cell lymphoma being evaluated for CAR-T therapy. Before treatment, a blood sample is analyzed by mass cytometry to assess the patient’s T cell profile. The analysis reveals a high proportion of stem cell memory T cells, which have been associated with better CAR-T outcomes. This information gives the oncologist confidence in proceeding with CAR-T therapy. During treatment, regular mass cytometry analyses track the expansion and persistence of CAR-T cells, allowing for real-time monitoring of treatment efficacy.
Mass cytometry has transformed our ability to monitor cancer treatment response and resistance, bringing unprecedented resolution to clinical decision-making. From predicting immunotherapy outcomes to uncovering hidden resistance mechanisms, this technology is bridging the gap between complex cellular data and actionable clinical insights. As mass cytometry becomes more integrated into routine clinical care, we move closer to the goal of truly personalized cancer treatment, where therapy can be dynamically adjusted based on each patient’s unique cellular response. The future of oncology is one where the microscopic changes in a tumor’s cellular composition directly inform macroscopic treatment decisions, promising better outcomes for cancer patients worldwide.
CAR T-cells are amazing. They're like tiny superheroes in our body, fighting cancer. I find them fascinating, and there's still so much to learn about them. I worked on CAR T-cells at Novartis. It's big science and big business rolled into one. Here's a funny thing: some of my closest friends work on CAR T-cells too. One makes them in the lab, another plans how to use them to help patients. What are the odds? It's a small world, especially when you're dealing with these microscopic cancer fighters. Who knew that these engineered cells would not only help fight disease, but also connect old friends? So here's to CAR T-cells - bringing people together while battling cancer. Now that's what I call a win-win!
Guillaume Beyrend