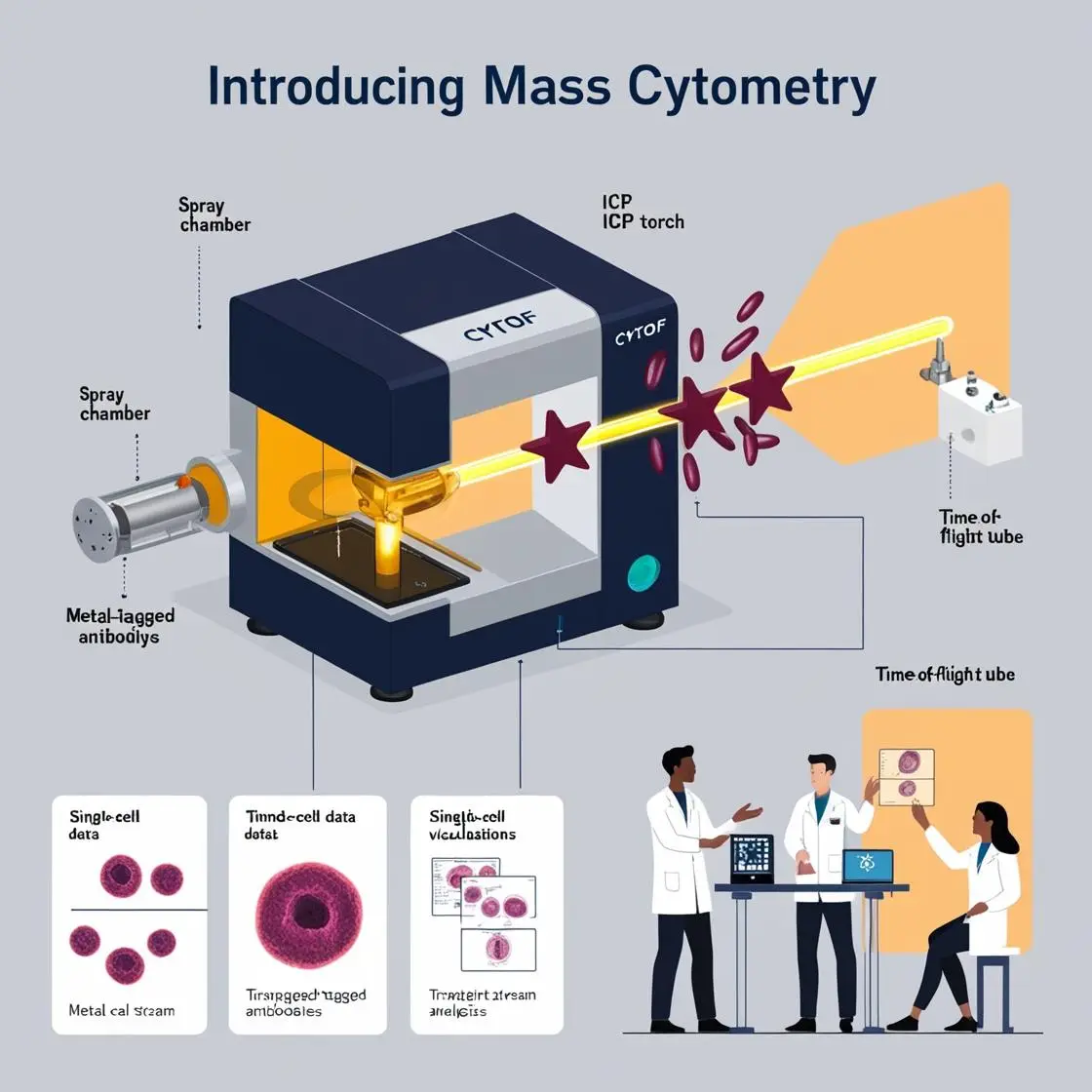
In mass cytometry, metal-conjugated antibodies are the secret agents of cellular interrogation. These clever molecular constructs allow us to peer into the inner workings of cells with unprecedented detail. Let’s dive into the fascinating world of metal labeling and antibody conjugation.
Chemistry of Metal Labeling
The magic of mass cytometry begins with the marriage of metals and antibodies. This union is not a simple affair but rather a carefully orchestrated chemical dance.
Lou et al.’s pioneering work (2008) laid the groundwork for polymer-based elemental tagging in their paper “Polymer-Based Elemental Tags for Sensitive Bioassays”. They introduced a method using metal-chelating polymers to attach multiple metal ions to a single antibody. This approach significantly amplified the signal, making detection more sensitive.
Fast-forward to more recent times, and we see refinements in this chemistry. For instance, Han et al. (2020), in their paper “Metal-Labeled Monoclonal Antibody Conjugates for Use in Mass Cytometry” (Nature Protocol), introduced improved metal chelators. The authors meticulously detail protocols for conjugating rare earth metals to antibodies, enabling the creation of high-dimensional panels with up to 50 parameters for single-cell analysis. They provide three optimized protocols for conjugating monoclonal IgG antibodies with 48 high-purity heavy-metal isotopes: (i) 38 isotopes of lanthanides, 2 isotopes of indium, and 1 isotope of yttrium; (ii) 6 isotopes of palladium; and (iii) 1 isotope of bismuth. I used the protocol for Bismuth conjugation in my own papers and it works very well.
Advantages and Limitations of Metal Tags
Advantages:
- Minimal Signal Overlap: Unlike fluorophores in flow cytometry, metal isotopes have minimal overlap in their mass peaks, allowing for more parameters to be measured simultaneously.
- Stability: Metal tags are not subject to photobleaching, a common issue with fluorescent dyes.
- Quantitative: The signal from metal tags is directly proportional to the number of tagged antibodies bound to the cell, providing more accurate quantification.
- Multiplexing Capacity: With over 50 detection channels available, mass cytometry offers unparalleled multiplexing capabilities.
Limitations:
- Sensitivity: While improving, metal-tagged antibodies are generally less sensitive than their fluorescent counterparts.
- Cell Viability: The cells are destroyed during analysis, preventing subsequent functional assays or cell sorting.
- Conjugate Stability: Metal ions can dissociate from the chelator over time, potentially leading to signal loss.
Strategies for Antibody Conjugation
The art of conjugating metals to antibodies has seen significant advancements. Here are some key strategies:
- Direct Conjugation involves attaching the metal-chelating polymer directly to the antibody. It’s straightforward but can sometimes affect antibody affinity. This technique is the one commercialized by Fluidigm (now Standard BioTools).
- Click Chemistry: A more recent approach, described by Boas et al. (2019) in “Click Chemistry-Based Method for the Easy Development of Mass Cytometry Reagents” (Scientific Reports, 2018, cited only 4 times), uses bio-orthogonal click reactions to attach metal tags. This method offers more control over the conjugation site and potentially better preservation of antibody function.
- Optimized protocols for conjugating monoclonal IgG antibodies : as already mentioned earlier.
Longevity of Conjugated Antibodies
An important practical consideration is how long these metal-conjugated antibodies remain effective. Surprisingly, there are limited published data on this topic, but I can share some insights from the field.
According to a technical note from Standard BioTools, the manufacturer of CyTOF systems, properly stored metal-conjugated antibodies can maintain their performance for up to a year. However, this can vary depending on the specific antibody and storage conditions.
A study by Schulz et al. (2019), “Stabilizing Antibody Cocktails for Mass Cytometry” (Cytometry Part A), provides some empirical data. They found that most metal-labeled antibodies maintained stable signal intensity for at least six months when stored at 4°C. Some antibodies even showed stable performance for up to 18 months.
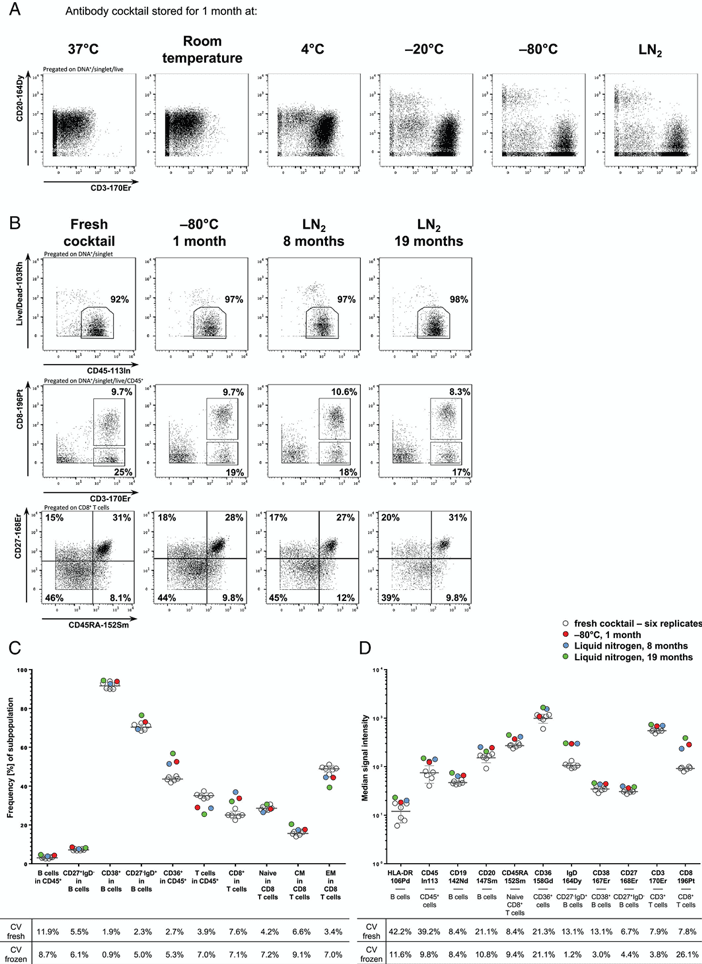
However, it’s important to note that stability can vary between different antibody clones and metal conjugates. Regular quality control checks are recommended, especially for critical experiments.
Metal-conjugated antibodies are the cornerstone of mass cytometry, enabling researchers to paint intricate molecular portraits of cells with unprecedented depth and precision. While they present their own unique challenges, the ongoing advancements in conjugation chemistry and stability continue to expand the horizons of high-dimensional single-cell analysis. As these molecular tools are refined and perfected, they bring us ever closer to unraveling the full complexity of cellular systems, promising new insights into the fundamental workings of life itself.
Picture this: there I was, cradling a tiny vial worth more than my entire grad school wardrobe. No, it wasn't liquid gold or a rare perfume – it was 3 mL of metal-conjugated antibodies, a cocktail so precious it made my hands shake like I'd had a few too many espressos. You see, in the world of mass cytometry, fresh is best. So, I decided to go big or go home – 200 samples big, to be exact. It was like preparing for a gourmet dinner party, but instead of farm-fresh vegetables, I was dealing with antibodies that cost more per milliliter than vintage champagne. The day of staining was like a scientific flash mob. Suddenly, the lab was swarming with 10 eager helpers, pipettes at the ready. It was a beautiful sight – a ballet of lab coats and precision timing. Who says scientists can't coordinate? Why all this fuss, you ask? Well, acquiring everything in one heroic week-long marathon meant the CyTOF's detector would have the same sensitivity throughout. It's like making sure the judge in a singing competition doesn't get a hearing aid halfway through – consistency is key! Sure, we could have spread it out over a year, but where's the fun in that? Plus, with the rate at which lab equipment gets upgraded, we might as well have been comparing apples to very expensive, highly sophisticated oranges. So there we were, a team of scientific commandos, armed with pipettes and a small fortune in antibodies, ready to stain our way into the annals of mass cytometry history. It was exhausting, exhilarating, and probably a bit mad – but hey, that's science for you. Just remember, the next time you see a beautifully consistent dataset, spare a thought for the bleary-eyed researcher who might have just pulled off the equivalent of a cytometric Olympics. Medal ceremony, anyone?
Guillaume Beyrend