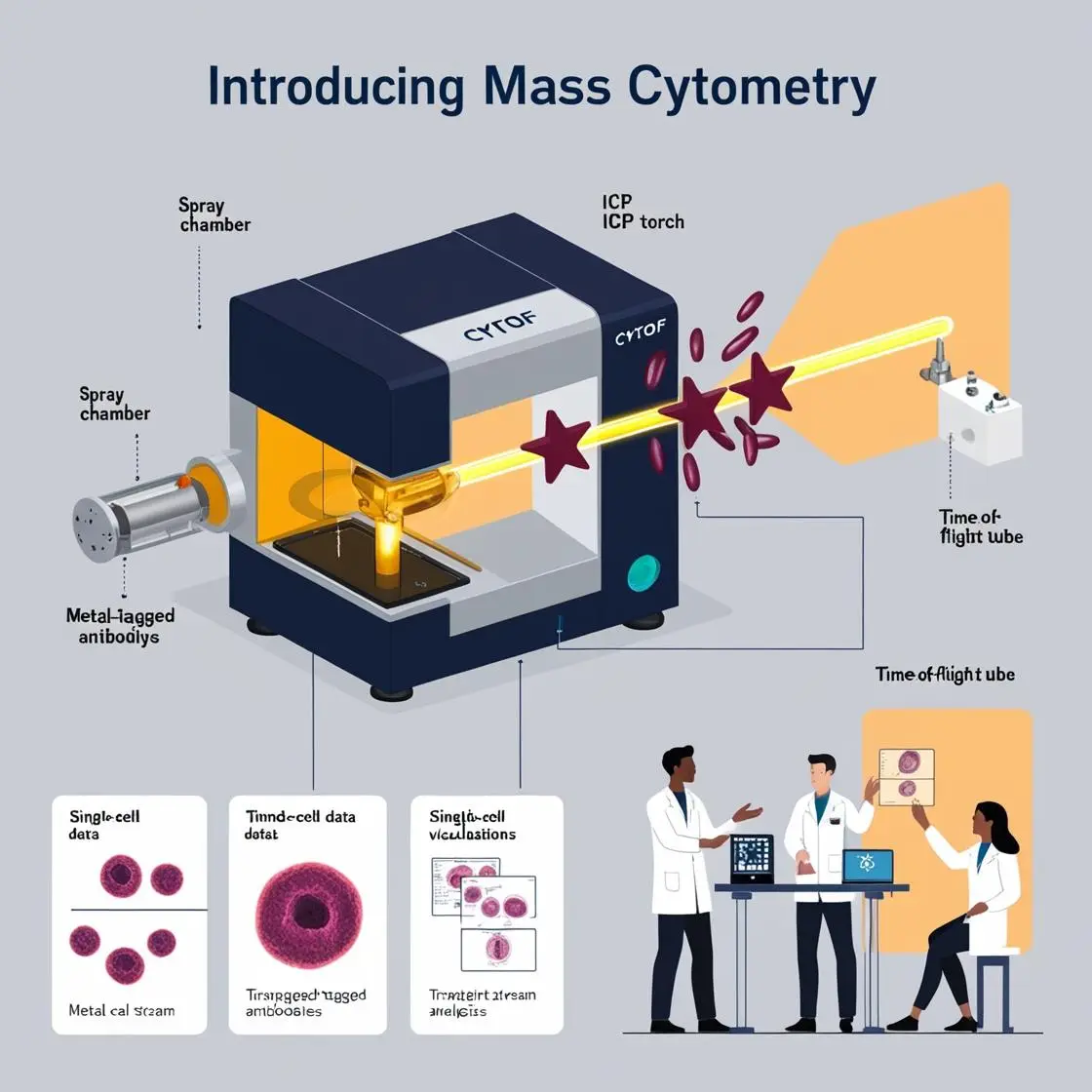
Metabolic disorders, including obesity and diabetes, have emerged as major global health challenges. Recent research has revealed a complex interplay between metabolism and the immune system, with mass cytometry (CyTOF) playing a crucial role in unraveling these intricate connections.
Immune Cell Metabolism in Obesity and Diabetes
The landmark study by Kratz et al. (2014) in Cell Metabolism, cited almost 800 times, laid the groundwork for understanding how metabolic dysfunction affects immune cell function, particularly in adipose tissue macrophages. Building on this foundation, recent CyTOF studies have provided even deeper insights into the immune-metabolic interface.
A pivotal study by Böhme et al. et al. (2020) published in Nature Communications, “Metformin enhances anti-mycobacterial responses by educating CD8+ T-cell immunometabolic circuits,” used CyTOF to show that metformin treatment expands a population of memory-like antigen-inexperienced CD8+CXCR3+ T cells in naive mice, and in healthy individuals and patients with T2D. This work demonstrated how metabolic interventions can have far-reaching effects on immune responses, potentially explaining some of the pleiotropic benefits of metformin.
Profiling Adipose Tissue Inflammation
CyTOF has been instrumental in characterizing the complex inflammatory milieu of adipose tissue in metabolic disorders.
A study by Jaitin et al. (2019) in Cell, “Lipid-Associated Macrophages Control Metabolic Homeostasis in a Trem2-Dependent Manner,” showed how sc-RNA-seq revealed a dynamic adipose tissue immune cell atlas in mice and humans. They identify an unique population of lipid-associated macrophages in adipose tissue. These cells play a crucial role in maintaining metabolic homeostasis, offering new targets for therapeutic intervention in obesity and diabetes.
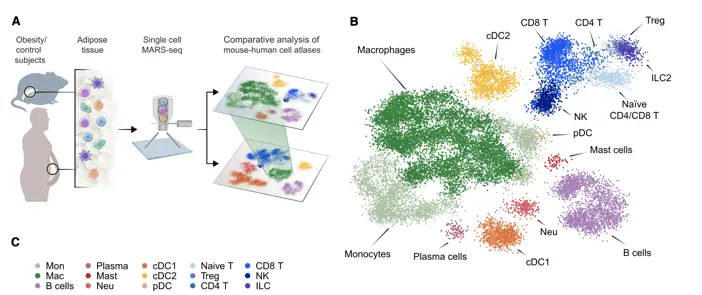
Biomarkers for Metabolic Syndrome
The high-dimensional capabilities of CyTOF have proven valuable in identifying novel biomarkers for metabolic syndrome.
Hildreth et al. (2020) in Nature Medicine, “Single cell sequencing of human white adipose tissue identifies novel cell states in health and obesity” used single cell technologies to create a comprehensive atlas of immune cell states in obesity. We report novel subsets and developmental trajectories of adipose-resident innate lymphoid cells (ILCs), dendritic cells (DCs) and monocyte-derived macrophage populations that accumulate in obese white adipose tissue.
The Metabolic Disorders-Cancer Connection
Emerging research has highlighted strong links between metabolic disorders and cancer risk. CyTOF has been instrumental in elucidating the mechanisms underlying this connection.
A groundbreaking study by Ringel et al. (2020) in Cell, cited over 425 times , “Obesity Shapes Metabolism in the Tumor Microenvironment to Suppress Anti-Tumor Immunity,” used sc-RNA-seq and flow cytometry to profile immune cells in the tumor microenvironment of obese versus lean mice. They found that obesity-induced metabolic changes suppress anti-tumor immunity, providing a mechanistic link between obesity and increased cancer risk. This work has been recently completed by the paper of Piening et al. in April 2024 “Obesity-related T cell dysfunction impairs immunosurveillance and increases cancer risk“, using similar techniques.
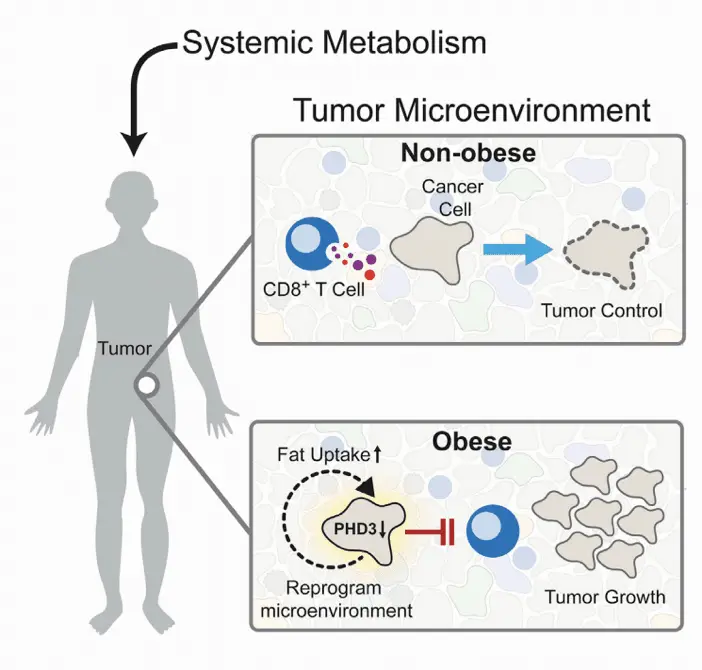
Furthermore, Elyada et al. (2021) in Cancer Discovery, “Cross-Species Single-Cell Analysis of Pancreatic Ductal Adenocarcinoma Reveals Antigen-Presenting Cancer-Associated Fibroblasts,” cited over 1400 times, employed single-cell RNA sequencing to characterize the tumor microenvironment in pancreatic cancer, a disease strongly linked to metabolic disorders. They identified a unique subset of cancer-associated fibroblasts that may contribute to immune suppression in the tumor.
Exercise and Metabolic Health: A CyTOF Perspective
The benefits of exercise on metabolic health are well-established, and single cell analysis has provided new insights into the underlying mechanisms.
Rocha et al. (2022) in Cell Metabolism, “A single-cell atlas of human skeletal muscle responses to exercise and inactivity”. Researchers employed single-cell RNA sequencing to investigate the effects of exercise on cellular composition and specific processes in human skeletal muscle before and after intense exercise. They identified fifteen clusters originating from six different cell populations. While most cell populations remained quantitatively stable after exercise, mesenchymal, endothelial, and myogenic cells showed significant transcriptional changes. Additionally, three subpopulations of myogenic cells were characterized based on maturation stages, and exercise accelerated myogenic progenitor cells’ maturation trajectory. These findings enhance our understanding of muscle adaptations triggered by physical activity, relevant to conditions like sarcopenia, cachexia, and glucose homeostasis.
The application of single cell technology to metabolic disorder research has revealed the intricate connections between metabolism, immunity, and disease. From uncovering novel immune cell populations in adipose tissue to elucidating the mechanisms linking obesity to cancer, sc-RAN-seq has provided unprecedented insights into the cellular basis of metabolic health and disease.
As we continue to harness the power of high-dimensional single-cell analysis, we move closer to a comprehensive understanding of metabolic disorders. This knowledge paves the way for more targeted and effective therapies, potentially revolutionizing the treatment of conditions like obesity, diabetes, and their associated complications, including cancer.
The story of metabolic disorders, once thought to be primarily about energy balance, has been rewritten as a complex tale of immune dysfunction, cellular adaptation, and systemic dysregulation. Through the lens of CyTOF, we're beginning to see how each cell type plays its part in this intricate metabolic orchestra. As we decode this cellular symphony, we edge closer to conducting it harmoniously, offering hope for millions affected by metabolic disorders worldwide.
Guillaume Beyrend