Imagine trying to understand a bustling city by only looking at satellite images. You might see the general layout, but you’d need to catch the intricate details of individual lives that make the city come alive. This is the revolution that single-cell analysis has brought to biology and medicine.
The Power of One
Importance of Single-Cell Resolution
1. Unveiling Hidden Diversity: Bulk analysis can mask rare cell types or states. Single-cell resolution is like having a high-powered microscope for cellular biology, revealing previously invisible cellular landscapes.
2. Capturing Dynamics: Cells are not static entities. They transition between states, respond to stimuli, and interact with their environment. Single-cell analysis allows us to capture these dynamic processes in action.
3. Understanding Development: From embryogenesis to tissue regeneration, single-cell analysis helps us understand how complex organisms arise from a single cell.
4. Decoding Disease: Many diseases, particularly cancer, are characterized by cellular heterogeneity. Single-cell analysis helps us understand this complexity, paving the way for more effective treatments.
Heterogeneity: The Spice of Cellular Life
Implications for Personalized Medicine
The insights gained from single-cell analysis are revolutionizing our approach to medicine. Here’s how:
1. Precision Diagnostics: By understanding the unique cellular composition of a patient’s tissue, we can make more accurate diagnoses. For instance, in cancer, single-cell analysis can reveal the presence of rare drug-resistant cells that might be missed by bulk sequencing.
2. Tailored Treatments: Understanding cellular heterogeneity allows for more targeted therapies. Instead of a one-size-fits-all approach, treatments can be designed to address the specific cellular makeup of a patient’s disease.
3. Monitoring Treatment Response: Single-cell analysis enables us to track how individual cells respond to treatment in real time, allowing for rapid adjustments to therapeutic strategies.
4. Predicting Disease Progression: By understanding the cellular trajectories and transitions, we can better predict how diseases might progress in individual patients.
Recent Advances and Cutting-Edge Algorithms
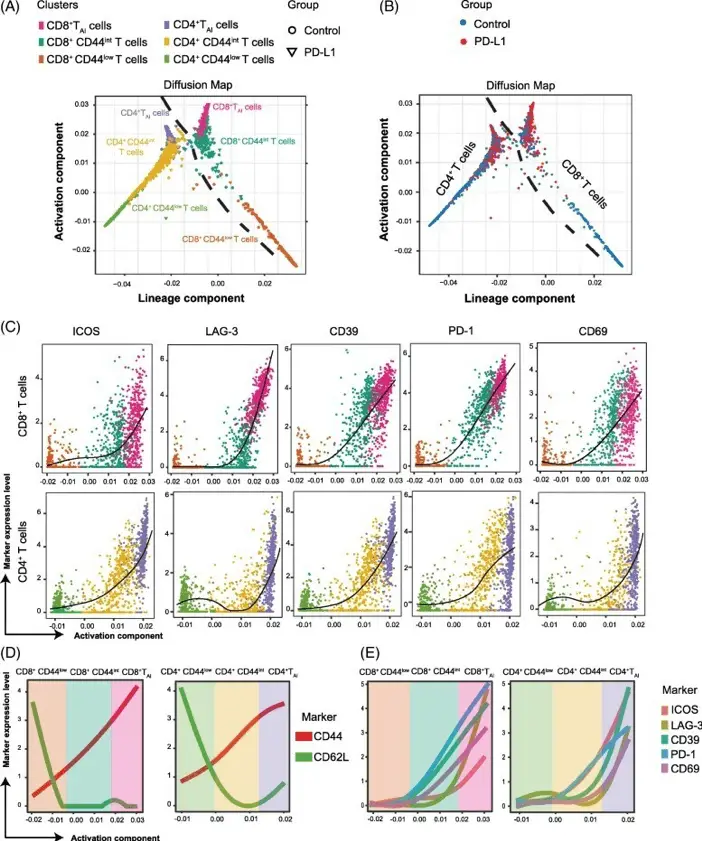
The field of single-cell analysis is rapidly evolving, with new algorithms and tools constantly emerging to tackle the complexities of cellular data. One such powerful tool is Destiny, an algorithm designed to study cell fate. Developed by Angerer, Haghverdi et al. and published in Bioinformatics in 2015,
Destiny uses diffusion maps to model the transitions between cell states, allowing researchers to trace developmental trajectories and predict cellular fates. This algorithm has been handy in understanding complex biological processes such as differentiation and disease progression at the single-cell level.
Destiny creates a diffusion map, representing the manifold of cell states. It calculates transition probabilities between cells based on their similarity, allowing for reconstructing continuous trajectories in high-dimensional space. This approach is compelling for analyzing developmental processes or other biological phenomena where cells undergo gradual changes in state.
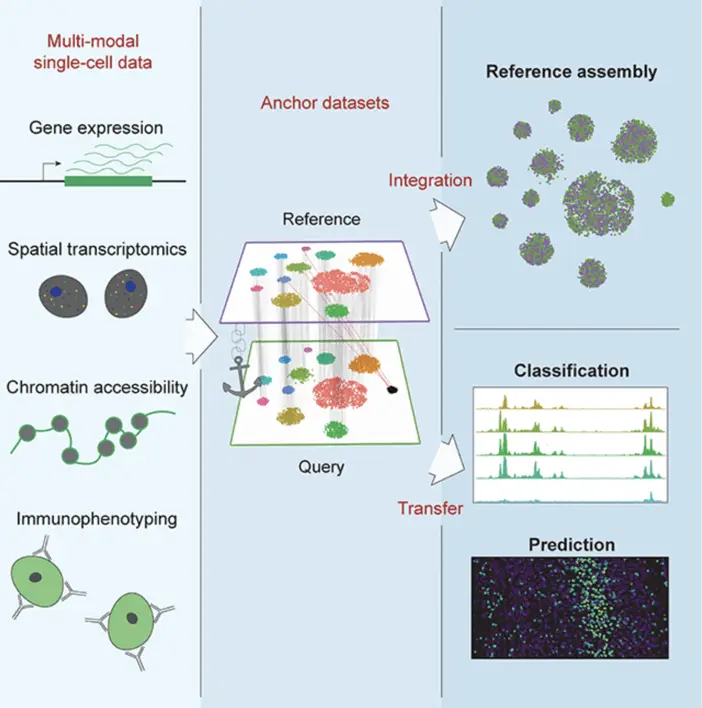
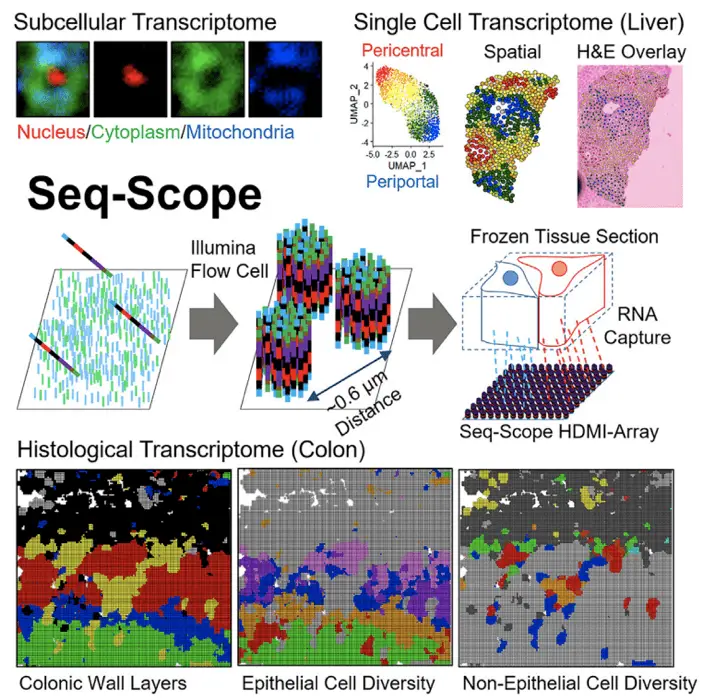
Another advancement is the integration of spatial information with single-cell transcriptomics. Methods like MERFISH (Multiplexed Error-Robust Fluorescence In Situ Hybridization), in Science, 2015, and Seq-Scope (Cell, 2021) are pushing the boundaries of what’s possible, allowing us to understand the molecular profiles of individual cells and their spatial context within tissues.
Machine learning and artificial intelligence are also increasingly important in single-cell analysis. Techniques like deep learning are being applied to automatically identify cell types, predict cell fates, and even generate hypothetical cell states that might exist under different conditions.
As Stuart and colleagues demonstrate in their 2021 Cell paper, the future of single-cell analysis lies in integration – bringing together different types of single-cell data to build a more comprehensive understanding of cellular systems. This integrative approach already yields insights into complex biological processes like embryonic development, tumor evolution, and immune system function.
Single-cell analysis represents a paradigm shift in our understanding of biology and medicine. It’s as if we’ve moved from viewing life through a kaleidoscope to seeing it through a high-definition microscope. As we refine our tools and analytical methods, we edge closer to unraveling the incredible complexity of life at its most fundamental unit – the single cell.
For students embarking on their scientific journey, single-cell analysis offers a frontier of discovery. It’s a field where biology meets big data, where each cell has a story to tell, and where the next big breakthrough in medicine might be hiding in the unique profile of a single cell.
Ah, Destiny. I used it in my paper in 2019. No, not the mystical force guiding our lives, but a cheeky little algorithm I once used that got me more attention than a puppy in a park. There I was, feeling like a digital Sherlock Holmes, unraveling the mysteries of biology with nothing but ones and zeros. That's when I realized I needed to be a scientific double agent - part code-slinging keyboard warrior, part pipette-addict. It's like being bilingual, but instead of French and English, you're fluent in R and Petri dish. Trust me, it's handier than a pocket-sized Swiss Army knife at a black-tie dinner. So there I was, straddling the divide between silicon and cells, trying not to pull a mental hamstring. But it works. Because in the end, science is all about bridging gaps - between theory and practice, between digital and biological. So here's to Destiny, to reducing dimensions without reducing our sense of wonder, and to remembering that sometimes, the best scientific instrument is a brain that can hop between wetware and software without getting soggy. Now, if you'll excuse me, I need to go explain to my computer why it can't wear a lab coat.
Dr. Guillaume Beyrend-Frizon