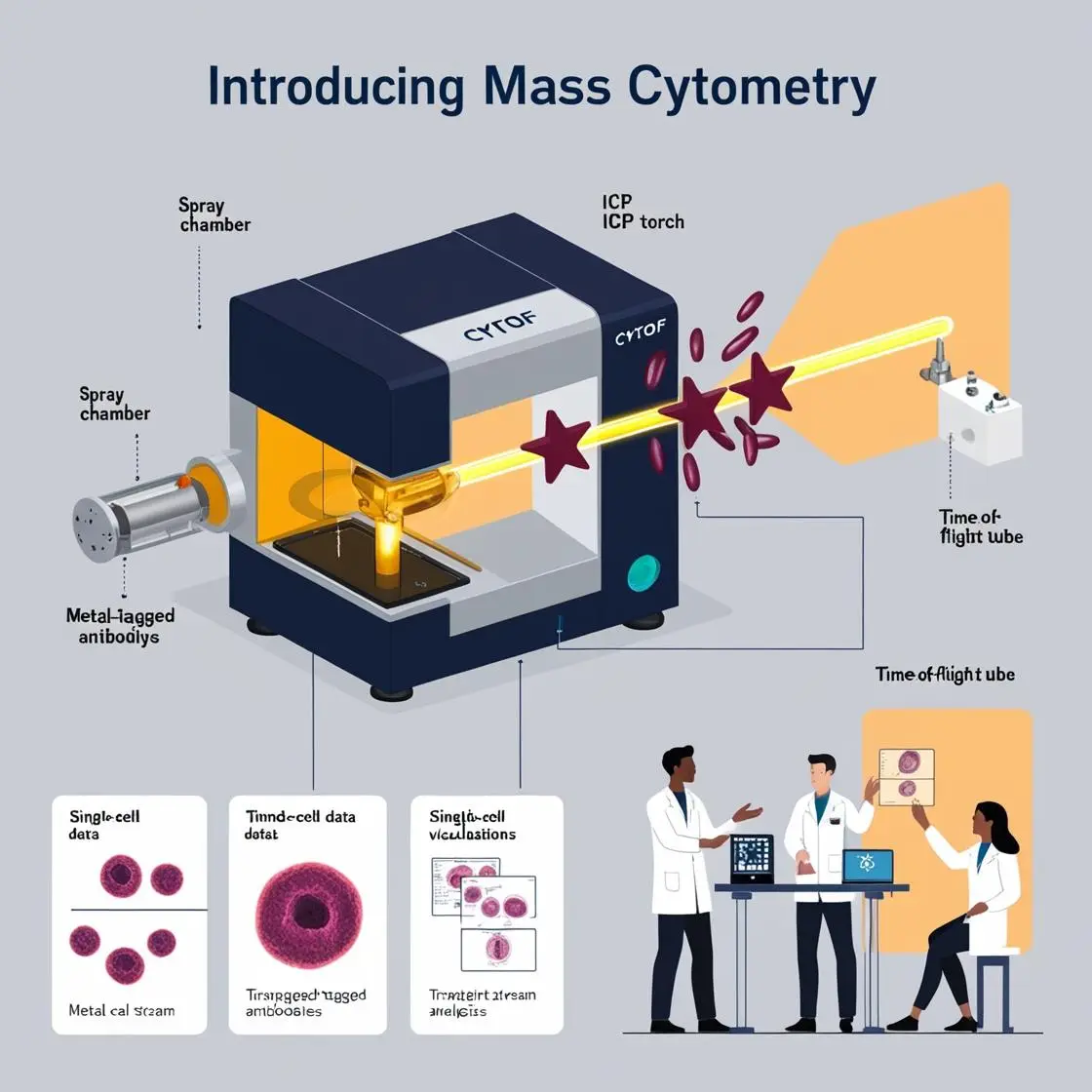
The field of infectious diseases has been significantly advanced by the application of mass cytometry (CyTOF), offering unprecedented insights into host-pathogen interactions, immune responses, and disease progression. This chapter focuses on how CyTOF has revolutionized our understanding of infectious diseases, with a particular emphasis on HIV/AIDS research.
Characterizing Pathogen-Specific T Cells
The ability to identify and characterize pathogen-specific T cells with high precision has been a game-changer in infectious disease research. CyTOF has been particularly powerful in this area.
HIV/AIDS Research: A CyTOF Revolution
While CyTOF has greatly advanced our understanding of HIV, some of the most profound insights have come from rare cases of HIV cure. The “Berlin Patient,” Timothy Ray Brown, became the first person cured of HIV in 2007. Brown, who was HIV-positive and diagnosed with acute myeloid leukemia, received a stem cell transplant from a donor with a rare genetic mutation (CCR5-delta32) that confers resistance to HIV infection.
Following Brown’s case, the “London Patient,” Adam Castillejo, became the second person cured of HIV in 2019 through a similar stem cell transplant procedure. CyTOF played a crucial role in monitoring these patients’ immune reconstitution and confirming the absence of replication-competent virus.

The Power of single-cell technologies in Unraveling HIV Mysteries
CyTOF ability has been instrumental in decoding the complex immune landscapes in HIV infection, treatment, and cure, as it has been recently described in a comprehensive review “Advances in HIV research using mass cytometry” by George et al. (2023).
- Viral Reservoir Characterization: single cell technologies have helped identify the specific cell subsets that harbor latent HIV, guiding strategies for targeted reservoir elimination, as statednrecently in a study (2023) by Clark et al. “HIV silencing and cell survival signatures in infected T cell reservoirs”,
- Immune Exhaustion Profiling: High-dimensional analysis has revealed the intricate patterns of T cell exhaustion in chronic HIV infection. Bengsch et al. (2018) in Immunity used CyTOF to characterize exhausted T cell subsets in HIV and other chronic viral infections, informing approaches to reinvigorate antiviral immunity.
- Immune Reconstitution Analysis: CyTOF has been crucial in understanding immune recovery after antiretroviral therapy. Several papers have analysed such immune response by using CyTOF, as Bekele et al. in 2019 published in JCI. Briefly, . the results revealed that CD4+ T cell clusters with high expression of CD27, CD28, CD127, and CD44, whose function involves T cell migration to inflamed tissues and survival, are more abundant in healthy controls and patients initiating anti retroviral therapy (ART) during the acute phase; on the contrary, CD4+ T cell clusters in patients initiating ART during the chronic phase had reduced expression of these markers.
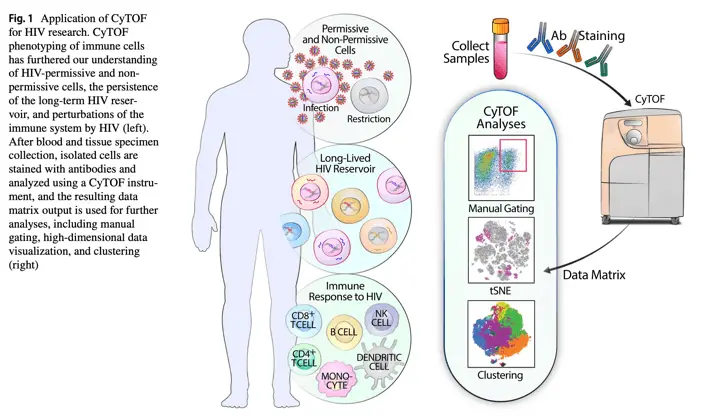
These studies demonstrate the significant impact CyTOF has had on our understanding of HIV pathogenesis, treatment responses, and potential cure strategies. By providing a high-resolution view of the immune landscape in HIV infection, CyTOF has opened new avenues for research and therapeutic development in the fight against this persistent viral foe.
The Journey Continues
As we reflect on the remarkable progress in HIV research, facilitated in no small part by CyTOF technology, we’re reminded of the power of persistence and innovation in tackling even the most formidable of diseases. From the early days of the AIDS crisis to the recent cases of HIV cure, each step forward has been hard-won through the dedication of researchers, clinicians, and patients alike.
The story of HIV research is far from over. Each CyTOF experiment, each data point, each newly identified cell subset brings us closer to unraveling the full complexity of HIV pathogenesis and immunity. As we peer through the high-dimensional lens of CyTOF, we’re not just seeing cells and markers – we’re witnessing the intricate dance of virus and host, played out on a cellular stage.
In the grand narrative of infectious disease research, CyTOF has opened a new chapter, allowing us to read the story of HIV and other pathogens with unprecedented clarity. As this technology continues to evolve, so too does our understanding of the delicate balance between humans and the microbes that challenge us.
The journey from the first identified AIDS cases to the recent instances of HIV cure spans decades of scientific endeavor. It’s a testament to human ingenuity and the relentless pursuit of knowledge. As we look to the future, armed with the powerful insights provided by CyTOF and inspired by the cases of cure we’ve witnessed, we carry with us a renewed sense of hope and purpose.
HIV fascinates me. Its complexity, its ability to target the immune system itself, and its rapid mutation rate make it a formidable foe. The fact that we're still struggling to develop an effective vaccine decades after its discovery is a testament to its cunning nature. Mass cytometry has become a powerful tool in HIV research. A search in PubMed reveals approximately 20 studies using CyTOF or similar technologies to investigate HIV as of 2023. This number, while significant, pales in comparison to the field of tumor immunology, where over 200 CyTOF or similar studies have been published. These CyTOF studies in HIV research have provided unprecedented insights into the virus's impact on the immune system. For instance, Cavrois et al. (2017) used mass cytometry to reveal how HIV remodels CD4+ T cells, while Bengsch et al. (2018) employed CyTOF to characterize exhausted T cells in HIV infection. Despite these advances, HIV continues to surprise us. Its ability to establish latent reservoirs and evade immune detection remains a significant challenge. But with each CyTOF study, we peel back another layer of HIV's complexity, inching closer to understanding this elusive virus. In the grand symphony of immunology, HIV plays a particularly intricate and challenging piece. Mass cytometry is helping us decipher its notes, still one cell at a time.
Guillaume Beyrend