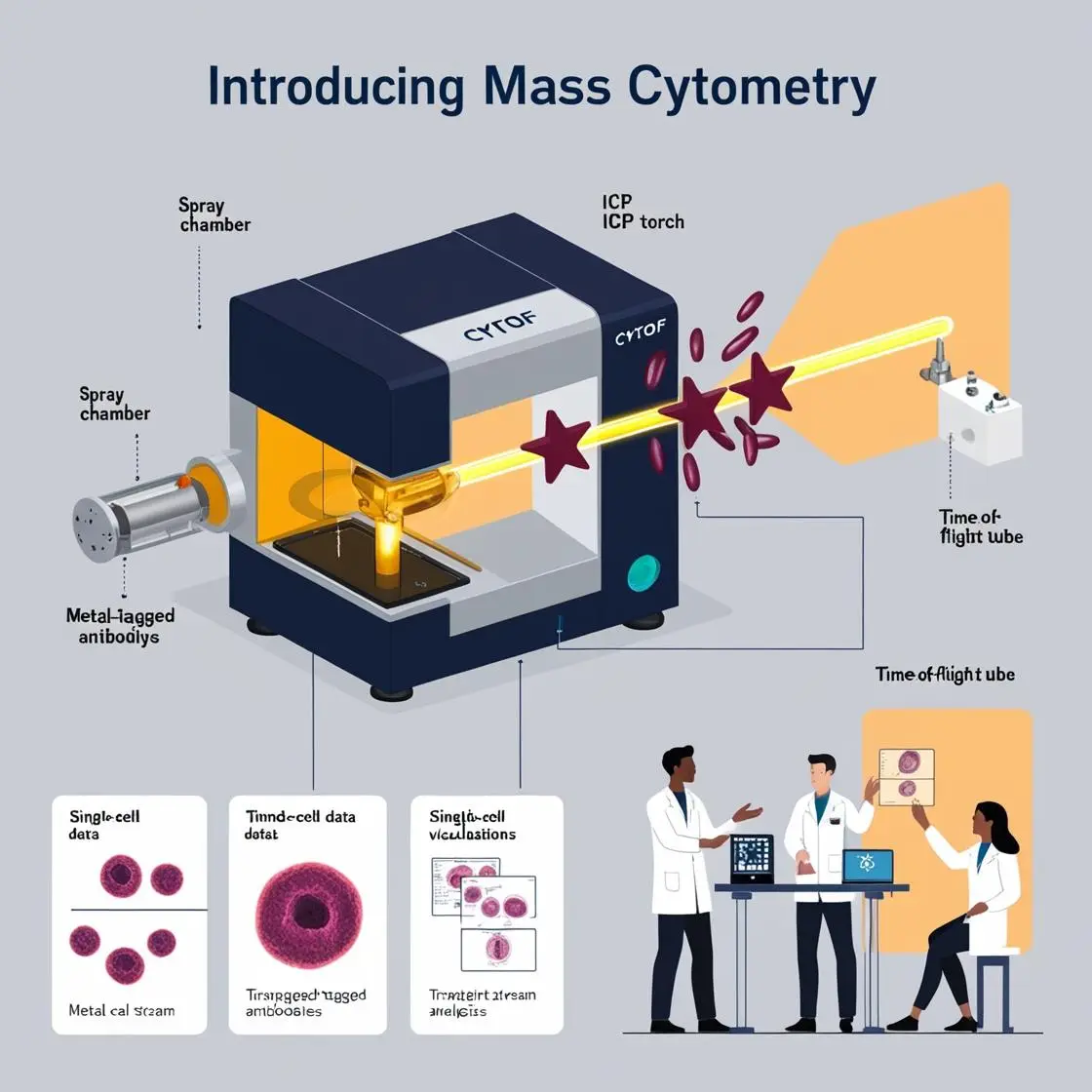
Hematological malignancies, encompassing a diverse range of leukemias and lymphomas, have been at the forefront of mass cytometry applications. The ability to analyze multiple parameters simultaneously at the single-cell level has revolutionized our understanding of these diseases, from classification to treatment monitoring. Let’s explore how mass cytometry is reshaping the landscape of hematological oncology.
Leukemia and Lymphoma Classification
Traditional classification methods for leukemias and lymphomas relied on a limited set of markers. Mass cytometry has dramatically expanded our ability to characterize these malignancies with unprecedented detail.
The landmark study by Amir et al. (2013) introduced viSNE, a tool for visualizing high-dimensional single-cell data. Applied to leukemia samples, viSNE revealed phenotypic heterogeneity that was previously undetectable, demonstrating the power of mass cytometry in refining disease classification. Thereafter, Amir launched his own data analysis company, Astrolabs.
A recent breakthrough came from Hansmann et al. (2022) in Nature Medicine, titled “Single-cell proteomic profiling identifies distinct tumor-associated macrophage subsets in diffuse large B cell lymphoma.” Using a 42-marker mass cytometry panel, they uncovered novel macrophage subsets with prognostic significance in diffuse large B cell lymphoma, potentially opening new avenues for targeted therapies.
Minimal Residual Disease Detection
One of the most impactful applications of mass cytometry in hematology has been in the detection of minimal residual disease (MRD) – the small number of cancer cells that may remain after treatment.
Astle et al. in Frontiers (2021) reviewed several mass cytometry studies and showed how markers are used for MRD detection in hematologic malignancies. This review emphasized superior sensitivity compared to traditional flow cytometry methods.
The enhanced sensitivity of mass cytometry for MRD detection is particularly crucial in diseases like acute lymphoblastic leukemia (ALL), where early identification of residual disease can guide treatment decisions and improve outcomes.
Prognostic Marker Identification
Mass cytometry has excelled in identifying novel prognostic markers in hematological malignancies. The high-dimensional data allows for the discovery of subtle cellular phenotypes that correlate with disease outcomes.
An example comes from Cader et al. (2018) in Blood, “Mass cytometry of Hodgkin lymphoma reveals a CD4+ regulatory T-cell-rich and exhausted T-effector microenvironment” This study used mass cytometry to uncover a specific T cell subset that might be linked to ineffective responses to immunotherapy in Hodgkin lymphoma patients.
Advantages of CyTOF in Hematology
Hematological malignancies are particularly well-suited for mass cytometry analysis for several reasons:
- Sample Accessibility: Blood and bone marrow samples are easily obtainable, providing a rich source of cells for analysis without the need for complex tissue processing.
- Single-Cell Suspension: Unlike solid tumors that require dissociation, blood samples are naturally in a single-cell suspension, ideal for mass cytometry analysis.
- Reduced Clogging Issues: The natural state of blood cells as individual entities reduces the risk of clogging in the CyTOF instrument, a common concern with other sample types.
- Established Marker Panels: The field of hematology has a long history of immunophenotyping, providing a strong foundation for designing comprehensive mass cytometry panels.
Recent Breakthroughs and Novel Markers
Mass cytometry has led to the discovery of several novel markers and cellular subsets in hematological malignancies:
- Levine et al. (2015) in Cell identified a previously unknown progenitor-like cell population in AML that correlated with poor prognosis.
- Bendall et al. (2019) in Cell used mass cytometry to create a single-cell atlas of human hematopoiesis, revealing new cellular intermediates and potential targets for leukemia therapy.
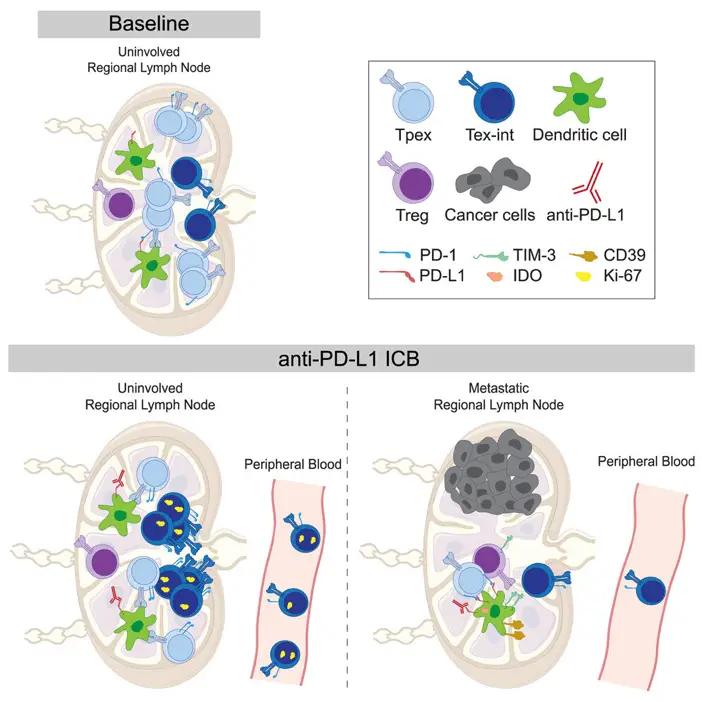
- A recent study by Rahim et al. (2023) in Cell, “Dynamic CD8+ T cell responses to cancer immunotherapy in human regional lymph nodes are disrupted in metastatic lymph nodes,” used mass cytometry to identify s the role of lymph nodes in human cancers.
4. « Mass Cytometry reveals unique phenotypic patterns associated with subclonal diversity and outcomes in multiple myeloma ». In this study published in Blood cancer Journal in May 2023, the researchers Baughn et al. identified 13 distinct phenotypic subpopulations across patients, some of which correlated with clinical outcomes. Notably, one subpopulation characterized by high CD45 and low BCL-2 expression was associated with better treatment response and survival.
The power of mass cytometry in hematological oncology cannot be overstated. From refining disease classification to uncovering novel prognostic markers and enhancing minimal residual disease detection, this technology has become an indispensable tool in the hematologist’s arsenal. The unique advantages of applying mass cytometry to blood-based malignancies have accelerated discoveries and are paving the way for more personalized and effective treatments. As researchers continue to push the boundaries of what’s possible with this technology, we stand at the threshold of a new era in hematological oncology. In this landscape, each patient’s disease can be characterized with exquisite detail, bringing us ever closer to the promise of truly personalized medicine. The symphony of cellular diversity in blood cancers, once muted, now resonates clearly through the lens of mass cytometry, composing a new understanding of these complex diseases.
Hematology and CyTOF? Now that's a match made in scientific heaven. It's like liquid biopsies were designed with CyTOF in mind - no clogging, no fuss, just cells living their best life in suspension. Sure, we can image lymph nodes on the Hyperion, but let's face it, blood samples are the smooth operators of the cellular world. They glide through the CyTOF like a hot knife through butter. But here's the kicker: we're sitting on a gold mine of hematology data, and we're barely scratching the surface. It's like having a library full of books and only reading the covers. Routine biopsies, Matutes scores, flow cytometry data - it's all there, begging to be recycled, reused, and reimagined through the lens of CyTOF. Imagine if we could turn every routine blood test into a treasure trove of research potential. It's not just possible - it's practically begging to happen. We just need to stop treating our data like a one-hit wonder and start seeing it as the gift that keeps on giving.
Guillaume Beyrend