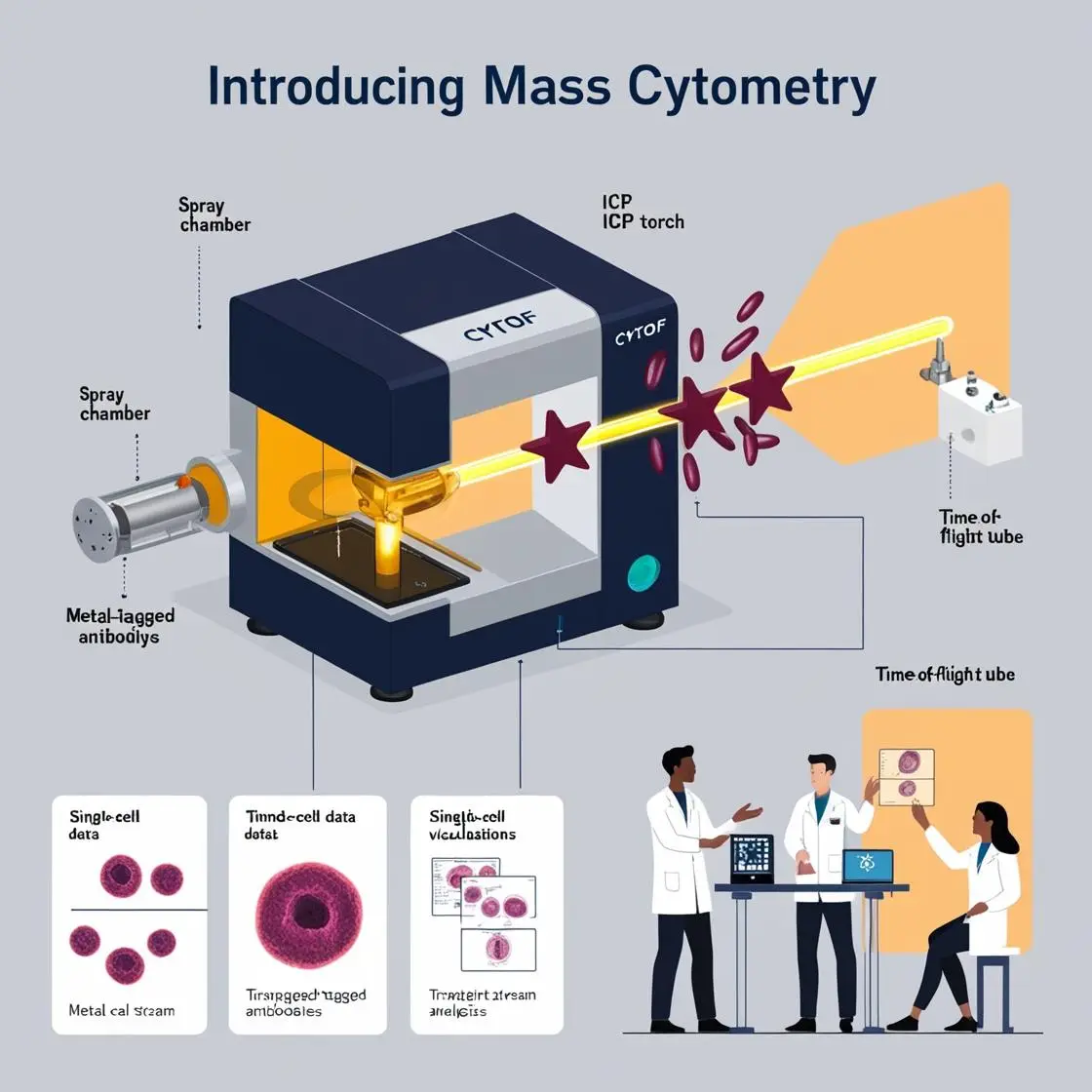
The intersection of cardiovascular disease and immunology might not seem obvious at first glance, but recent research has revealed a complex interplay between the immune system and heart health. Mass cytometry (CyTOF) has emerged as a powerful tool in unraveling this intricate relationship, offering new insights into atherosclerosis, heart failure, and myocardial infarction recovery.
Atherosclerosis and Plaque Composition
Atherosclerosis, once thought to be primarily a lipid storage disease, is now recognized as a chronic inflammatory condition. CyTOF has been instrumental in characterizing the diverse immune cell populations within atherosclerotic plaques.
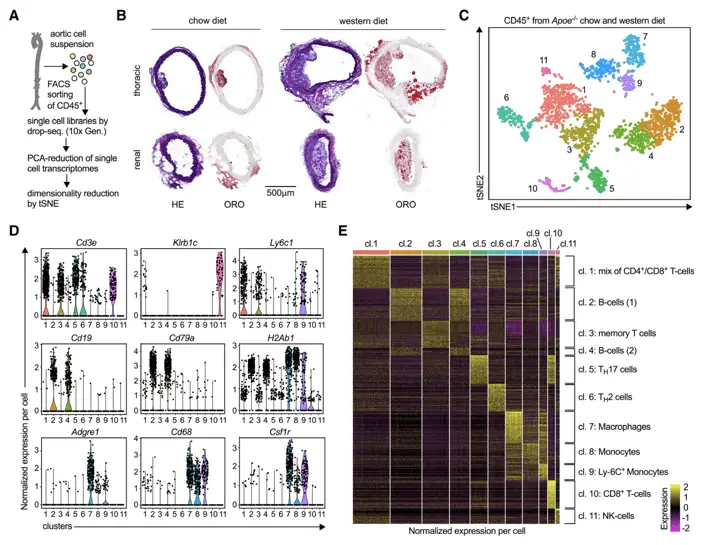
Winkels et al. (2018) published a groundbreaking study cited over 465 times in Circulation Research, creating an atlas of immune cell repertoire in mouse atherosclerosis using single-cell RNA-sequencing and mass cytometry. This work revealed unprecedented detail in the composition of atherosclerotic plaques, identifying novel immune cell subsets potentially involved in disease progression.
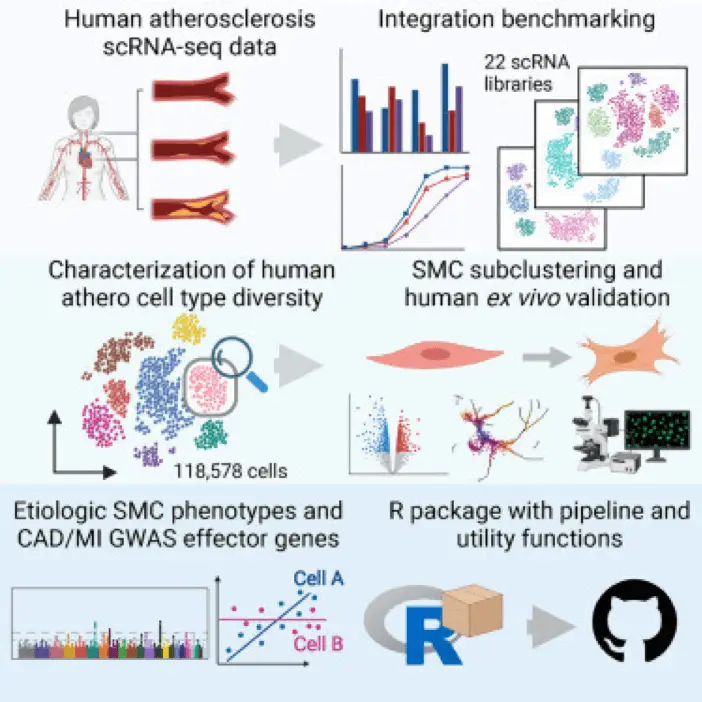
Building on this foundation, a recent study by Fernandez et al. (2023) in Cell Reports, “Integrative single-cell meta-analysis reveals disease-relevant vascular cell states and markers in human atherosclerosis,” used single-cell RNA-seq to profile human atherosclerotic plaques. They identified a specific pro-inflammatory macrophage population associated with plaque instability, providing a potential target for therapeutic intervention.
Immune Cell Involvement in Heart Failure
The role of immune cells in heart failure has been increasingly recognized, and CyTOF has provided crucial insights into this complex pathology.
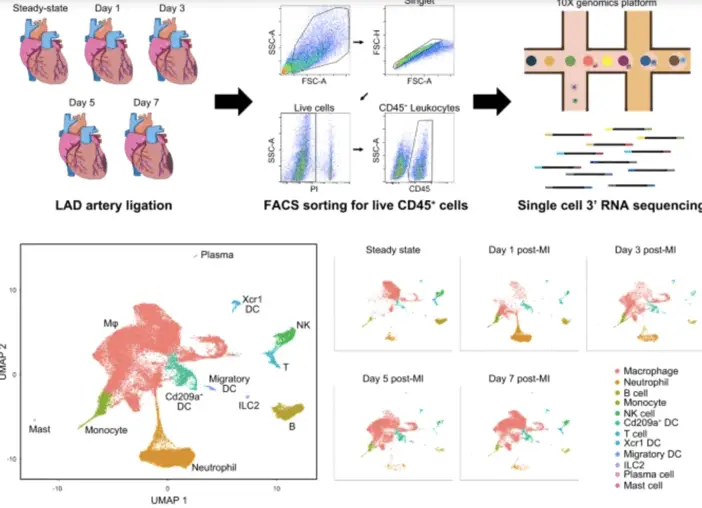
A recent study by de Winter et al. (July 2024) in Circulation, “Profiling cardiac catecholamine-producing cells after myocardial infarction by single-cell RNA sequencing and mass cytometry,” re-used flow cytometry and publicly available datasets to characterize immune cell populations in the heart after myocardial infarction. They discovered that a healthy heart contains diverse B and T cell populations, including effector subsets, which are regulated by homeostatic transcription factors. Following a myocardial infarction (MI), these cells are rapidly activated and contribute to acute wound healing, but they also maintain changes in their regulatory mechanisms, resulting in persistent pro-inflammatory activation. Analysis of single-cell RNA sequencing data from mouse and human hearts reveals that post-MI T and B cells upregulate pro-inflammatory and fibrotic genes, and maintain elevated activity in transcription factors related to inflammation and immunoglobulin production, suggesting their involvement in both acute healing and chronic immunopathology after MI.
This work is in continuation with previous study from Koening et al. (2022) published a study in Nature Cardiovascular Research, “Single-cell transcriptomics reveals cell-type-specific diversification in human heart failure” Using single-cell RNA sequencing, researchers define the cell composition of the healthy and failing human heart. They identify cell-specific transcriptional signatures associated with age and heart failure and reveal the emergence of disease-associated cell states. Notably, cardiomyocytes converge toward common disease-associated cell states, whereas fibroblasts and myeloid cells undergo dramatic diversification.
Monitoring Cellular Changes after Myocardial Infarction
Single cell RNA-sequencing has proven invaluable in tracking the dynamic cellular changes that occur following myocardial infarction. It has revolutionized our ability to monitor cellular changes after myocardial infarction (MI) with unprecedented resolution. In their study “Spatiotemporal dynamics of macrophage heterogeneity and a potential function of Trem2hi macrophages in infarcted hearts“, Jung et al. (2022) performed scRNA-seq on CD45+ leukocytes isolated from mouse hearts at various time points following MI. This technique allowed for the identification of 12 broad cell types and multiple subtypes, revealing the complex heterogeneity of immune cells in the infarcted heart. To complement the scRNA-seq data, the researchers employed spatial transcriptome sequencing (ST-seq) using the Visium Spatial Gene Expression platform, which provided spatial context to the gene expression profiles. The integration of scRNA-seq and ST-seq data, facilitated by bioinformatics tools such as Seurat and SPOTlight, enabled a comprehensive understanding of both the transcriptional states and spatial distribution of immune cells following MI. This approach revealed the dynamic changes in macrophage subpopulations over time, including the identification of a Trem2-high macrophage subset in the late phase of MI. The combination of these advanced sequencing technologies provides a powerful toolkit for elucidating the intricate cellular dynamics in the heart post-MI, potentially guiding the development of targeted therapies for improved cardiac recovery.
The CyTOF Twist: Marker Discovery and Beyond
While the connection between cardiovascular disease and immunology might not be immediately apparent, CyTOF has played a crucial role in bridging this gap. Here’s where the story takes an interesting twist:
- Revealing the “Immune Landscape” of the Heart: In their review in Nature Reviews Caridology, Fernandez et al. (2021) discuss how single-cell technologies such as single-cell mass cytometry, single-cell RNA sequencing and cellular indexing of transcriptomes and epitopes by sequencing are ideal for mapping the cellular and molecular composition of human atherosclerotic plaques and how these data can aid in the discovery of new precise immunotherapies.
- Personalized Cardiovascular Medicine and Bridging Cardiology and Immunology: The detailed immune profiling enabled by single cell sequencing opens the door to personalized treatment approaches. Depuydt et al. (2023) in Nature Cardiovascular Research demonstrated how single-cell T cell receptor seqencing analysis of atherosclerotic plaques could guide targeted immunomodulatory therapies by suggesting that atherosclerosis has an autoimmune compondent driven by autoreactive CD4+ T cells.
The application of CyTOF to cardiovascular research has revealed that the heart, far from being isolated from the immune system, is intimately connected to it. This realization is reshaping our approach to cardiovascular diseases, from prevention to treatment.
As we continue to explore the intricate dance between the heart and the immune system through the lens of CyTOF, we move closer to a future where cardiovascular diseases are not just treated, but potentially prevented or reversed through immune modulation. The beat of a healthy heart, we now understand, is conducted in concert with a finely tuned immune orchestra, and CyTOF is helping us decode this complex symphony, one cell at a time.
Guillaume Beyrend