The Dawn of Mass Cytometry in Biomarker Discovery
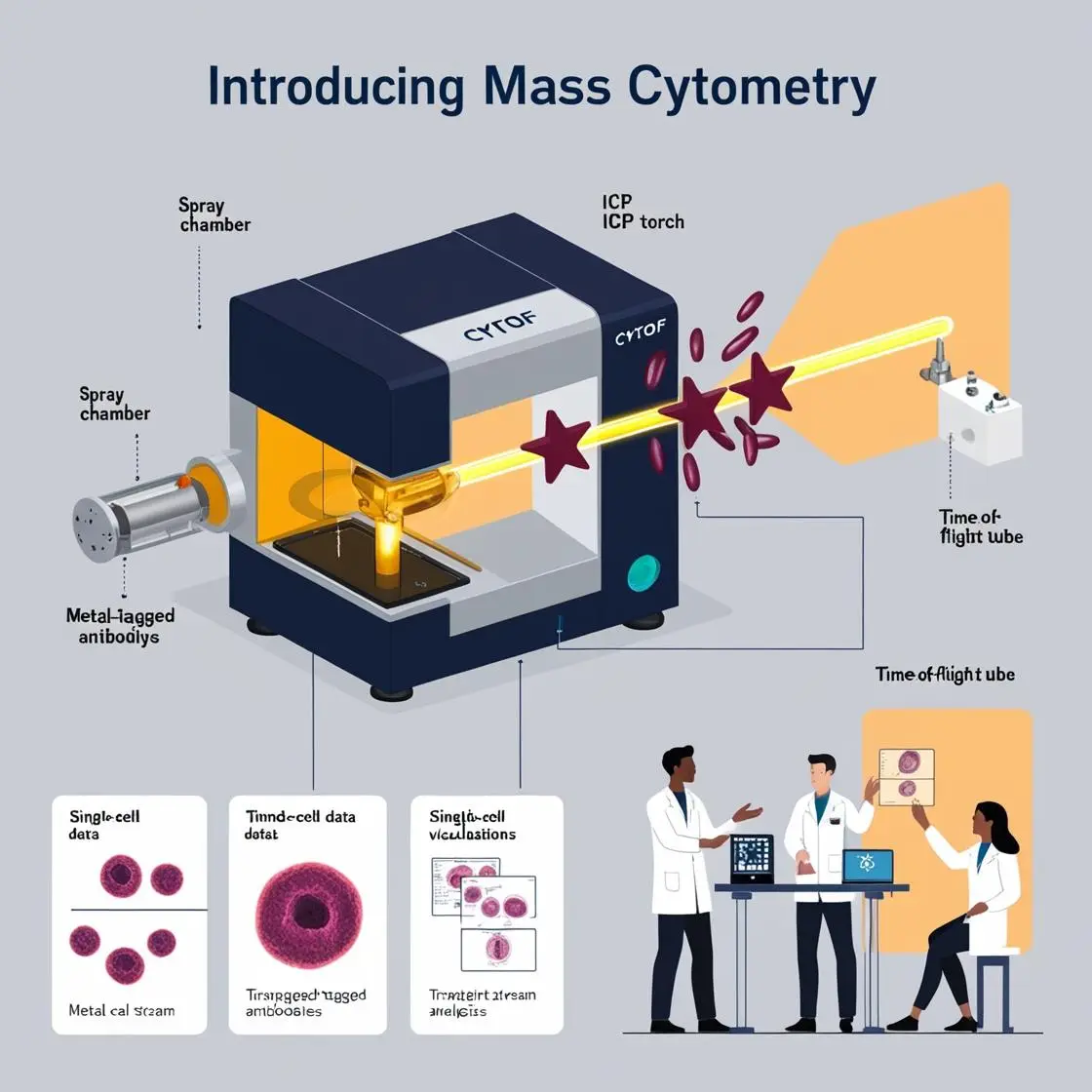
The groundbreaking work of Bendall et al., published in Science in 2011 (cited over 2000 times), marked a pivotal moment in the field of biomarker discovery. Their study, “Single-cell mass cytometry of differential immune and drug responses across a human hematopoietic continuum,” demonstrated the unprecedented power of mass cytometry to analyze multiple parameters at the single-cell level simultaneously. They specifically used mass cytometry, not flow cytometry. This study was, in fact, one of the pioneering works that introduced mass cytometry (CyTOF – Cytometry by Time of Flight) as a powerful tool for high-dimensional single-cell analysis.
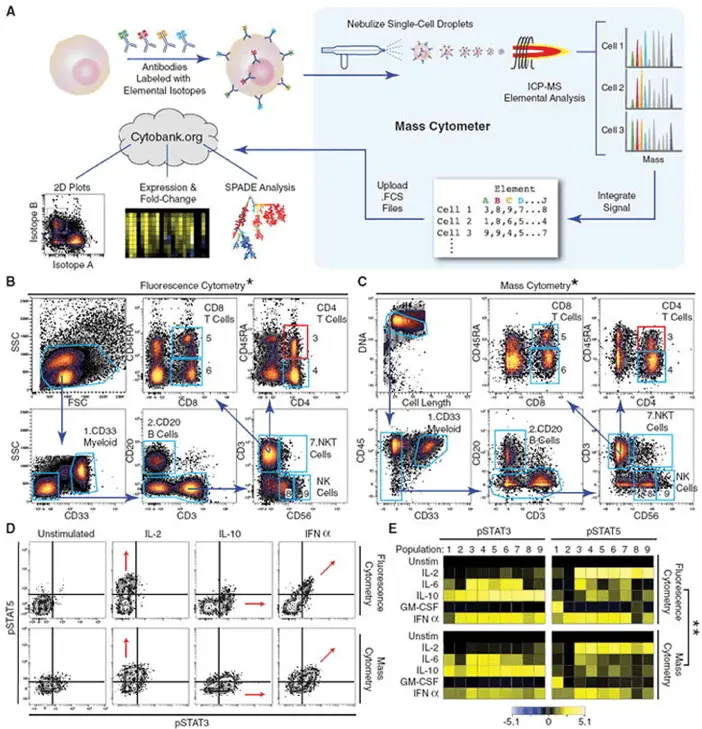
Using a panel of 31 antibodies, Bendall and colleagues (in Nolan lab, we’ll talk about him later) were able to dissect the human hematopoietic system with extraordinary resolution. They identified previously unrecognized cell subsets and mapped cellular signaling responses to various stimuli. This work laid the foundation for using mass cytometry as a powerful tool for biomarker discovery, showcasing its ability to reveal complex cellular hierarchies and functional responses that were previously invisible to researchers.
The authors used a mass cytometry approach with 31 antibodies labeled with isotopically pure rare earth elements. This allowed them to simultaneously measure 34 parameters in single cells, a significant advancement over traditional flow cytometry capabilities.
The Role of Mass Cytometry in Biomarker Identification
Mass cytometry offers several critical advantages in biomarker discovery:
- High-dimensional analysis: Mass cytometry can uncover complex biomarker signatures that lower-dimensional techniques might miss by measuring dozens of parameters per cell.
- Single-cell resolution: This identifies rare cell populations that may harbor essential biomarkers.
- Protein-level analysis: Unlike genomic or transcriptomic approaches, mass cytometry directly measures protein expression and modification, providing a more immediate picture of cellular function.
- Flexibility: Mass cytometry panels can be easily customized to focus on specific biological pathways or cell types of interest.
Modern Advances: A Case Study in Biomarker Discovery
Just a few years after the Bendall study, in 2016, mass cytometry panel has already been extended to 45 markers. A prime example is the work of Hartmann et al., published in JEM, titled “High-dimensional single-cell analysis reveals the immune signature of narcolepsy.” This study used mass cytometry to analyze immune cells from narcolepsy patients and healthy controls. They employed a 45-marker panel to comprehensively characterize the immune system, far surpassing the capabilities available to Bendall et al. in 2011.
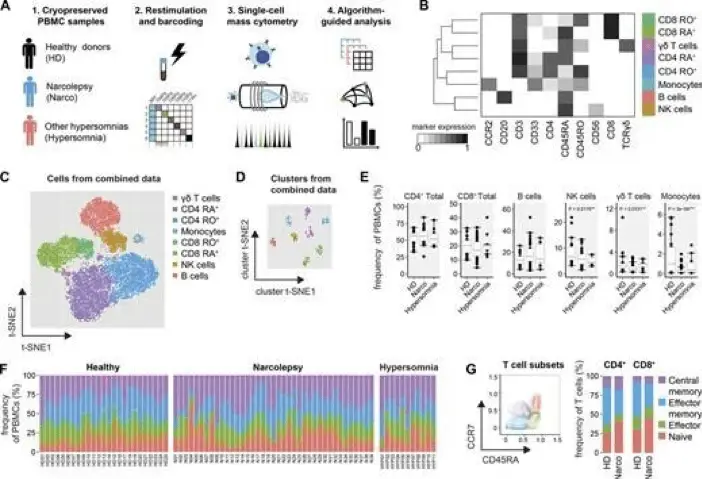
Hartmann and colleagues identified a subset of CD4+ and CD8+ T cells that was expanded in narcolepsy patients. This cell population, characterized by a unique combination of surface markers and functional properties, represents a potential biomarker for narcolepsy. Furthermore, their analysis revealed alterations in multiple immune cell types, painting a complex picture of the immune dysregulation associated with the disease.
This study exemplifies how modern mass cytometry approaches can:
- Identify complex biomarker signatures involving multiple cell types and markers.
- Reveal disease-associated changes in rare cell populations.
- Provide insights into disease mechanisms, potentially guiding therapeutic strategies.
Validation Strategies for New Biomarkers
Discovering a potential biomarker is only the first step. Rigorous validation is crucial before a biomarker can be used in clinical practice. Some key validation strategies include:
- Replication in independent cohorts: Findings must be reproducible in different patient populations.
- Orthogonal validation: Using complementary techniques (e.g., flow cytometry, immunohistochemistry) to confirm mass cytometry findings.
- Functional studies: Investigating the biological relevance of the identified biomarkers.
- Longitudinal studies: Assessing how biomarkers change over time or in response to treatment.
- Clinical correlation: Evaluating how well the biomarker correlates with clinical outcomes.
The journey from biomarker discovery to clinical application is long and complex, but mass cytometry has dramatically accelerated this process. Providing a high-dimensional, single-cell view of biological systems allows researchers to uncover unprecedented sophistication and specificity biomarkers.
As we look to the future, integrating mass cytometry with other high-dimensional techniques, such as single-cell transcriptomics and proteomics, promises to enhance further our ability to discover and validate biomarkers. This multimodal approach may ultimately lead to the development of exact, personalized diagnostic and therapeutic strategies, bringing us closer to the goal of truly personalized medicine.
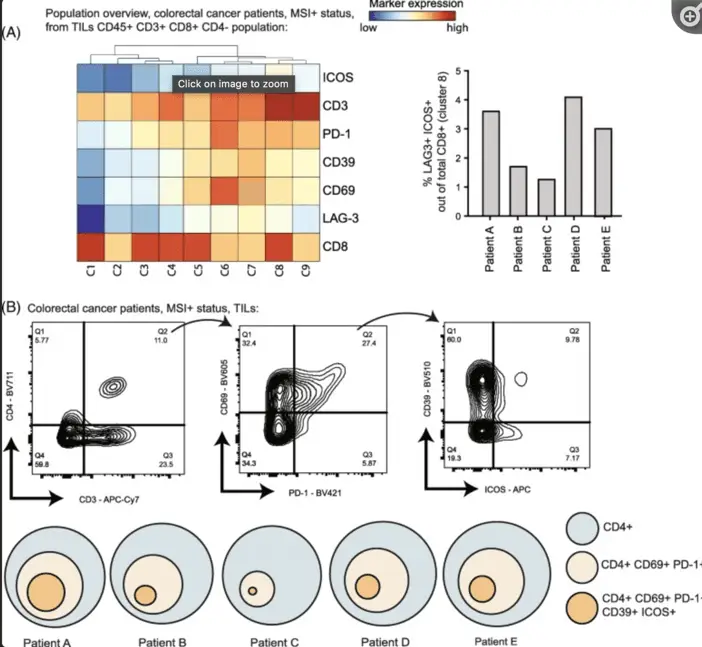
You know how some people fall in love at first sight? Well, I fell in love at first graph. There I was, swimming in a sea of data, when two little markers caught my eye: ICOS and LAG3. Suddenly, I was smitten. I started seeing them everywhere, like a lovesick teenager doodling names in a notebook. Before I knew it, I was introducing these markers to every lab mouse I could find. It was like setting up blind dates, but with antibodies and rodents.
Just as I thought I'd discovered the scientific equivalent of sliced bread, I learned that human trials were already happening. Talk about a reality check! It was like thinking you've invented the wheel, only to find out your neighbor's been running a bicycle shop.
But you know what? Seeing these antibodies actually help fight cancer was better than finding the toy in a cereal box. And to think, it all started with some squiggly lines on a computer screen. Who says science can't be romantic? Just remember, in research, sometimes love at first sight can lead to a breakthrough – or at least a really interesting lab report.Dr. Guillaume Beyrend-Frizon