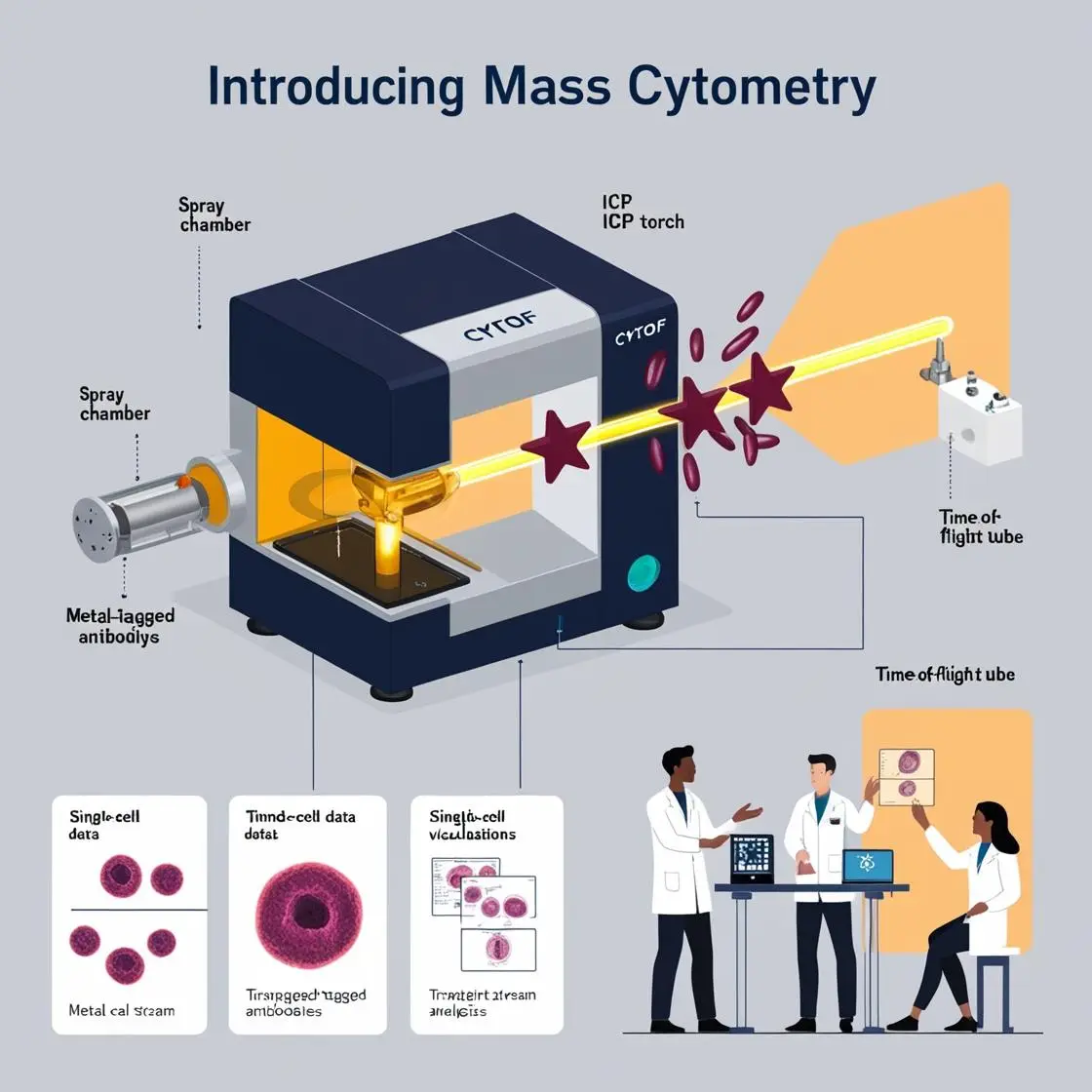
The convergence of artificial intelligence (AI) and mass cytometry has ushered in a new era of data analysis and interpretation in biomedical research and clinical practice. This powerful combination is revolutionizing our approach to understanding complex cellular systems and tailoring treatments for individual patients.
Deep Learning for Automated Cell Classification
One of the most impactful applications of AI in mass cytometry is automated cell classification. Traditional manual gating of cytometry data is time-consuming and subject to human bias. Deep learning algorithms have shown remarkable accuracy in identifying and classifying cell populations from high-dimensional cytometry data.
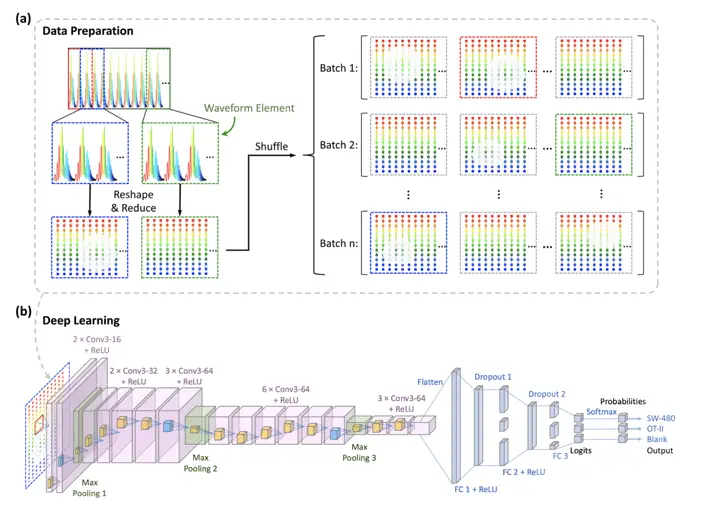
A landmark study by Li et al. (2019) published in Nature Methods, “Deep Cytometry: Deep learning with real-time inference in cell sorting and flow cytometry,” demonstrated how convolutional neural networks could be used for real-time cell classification in flow cytometry. While this study focused on flow cytometry, the principles have been successfully applied to mass cytometry data.
This paper introduces “deep cytometry,” a novel approach to cell analysis and sorting that combines label-free imaging with deep learning. Unlike traditional mass cytometry or flow cytometry, this method doesn’t require antibodies or any labeling. Instead, it uses time-stretch quantitative phase imaging (TS-QPI) to capture detailed information about cells’ physical properties as they flow through a microfluidic channel. A specialized laser and optical setup generate raw waveform data representing how light interacts with each cell. This data is then directly processed by a deep convolutional neural network, which can classify cells in real-time without the need for manual feature extraction or image conversion. The system achieved over 95% accuracy in distinguishing between cancer cells and white blood cells, and can make classification decisions within milliseconds, enabling real-time cell sorting. While it doesn’t provide the protein-specific information of mass cytometry or the dimensionality reduction of techniques like UMAP, this approach offers a complementary tool for rapid, label-free cell analysis based on physical and morphological properties.
Predictive Modeling Using Mass Cytometry Data
The integration of AI with mass cytometry data has opened new avenues for predictive modeling in both research and clinical settings.
Shalek and Benson (2017) in Science Translational Medicine highlighted the potential of single-cell analyses, including mass cytometry, combined with machine learning for tailoring treatments. They discussed how these approaches could lead to more personalized therapeutic strategies in various diseases.
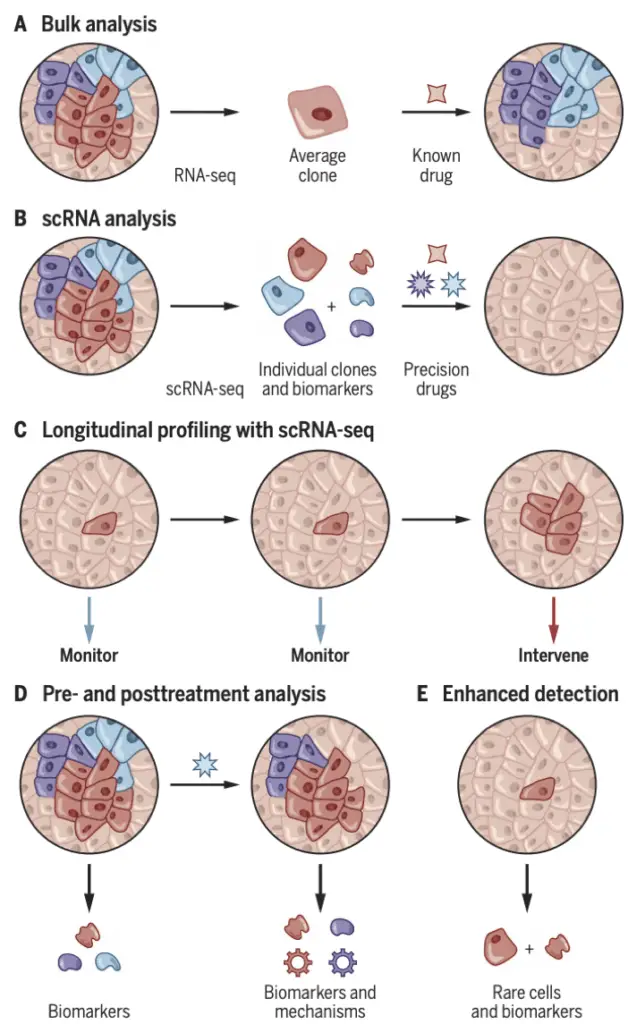
As we continue to navigate this exciting intersection of AI and single-cell biology, we find ourselves on the cusp of a new era in precision medicine. Each dataset analyzed, each algorithm developed, brings us closer to a future where treatments are truly personalized, guided by an AI-enhanced understanding of each patient’s unique cellular landscape. The symphony of cells that composes our biology is complex, but with AI as our interpreter, we are beginning to understand its intricate melodies and harmonies in ways previously unimaginable. This convergence of technologies is not just advancing our scientific understanding; it’s paving the way for more precise, effective, and personalized medical interventions, promising a future where each patient’s treatment is as unique as their cellular composition.
When I was developing Cytofast, I felt like a detective in a mystery novel where all the clues were written in a language I was still learning. Every tiny bug in the script sent me on a wild goose chase across the vast expanse of the internet. If only I had an AI sidekick back then! I could have saved thousands of hours and probably a few grey hairs. Now, with AI at our fingertips, it's like having a supercomputer in your pocket. It's amazing, sure, but you can miss the thrill of those early challenges. It's like the difference between exploring uncharted territory with just a compass and exploring with GPS - both get you there, but the stories you tell are quite different.
Guillaume Beyrend