Autoimmune diseases present a complex challenge in modern medicine, with the body’s defense system mistakenly attacking its own tissues. Mass cytometry has emerged as a powerful tool to decode these intricate disease mechanisms. Let’s explore how this technology has advanced our understanding of specific autoimmune conditions and their potential treatments.
Rheumatoid Arthritis: Unveiling Novel Cell Subsets
Rheumatoid arthritis (RA) has been a prime target for mass cytometry research, leading to groundbreaking discoveries.
The landmark study by Rao et al. (2017) published in Nature, cited almost 1000 times, “Pathologically expanded peripheral T helper cell subset drives B cells in rheumatoid arthritis,” used mass cytometry to identify a novel subset of T helper cells in RA patients. This PD-1hiCXCR5- T peripheral helper (Tph) cell population was found to promote B cell responses and inflammation. This discovery has fundamentally changed our understanding of RA pathogenesis, suggesting that these Tph cells, rather than follicular helper T cells, may be the primary drivers of B cell responses in RA.
Systemic Lupus Erythematosus: Mapping Cytokine Networks
In systemic lupus erythematosus (SLE), mass cytometry has been instrumental in mapping complex cytokine networks.
A study by Arazi et al. (2019) published in Nature Immunology, titled “The immune cell landscape in kidneys of patients with lupus nephritis,” used mass cytometry to analyze tissue-resident immune cells in an autoimmune condition. The researchers employed a 35-marker mass cytometry panel to profile kidney samples from patients with lupus nephritis, a severe complication of systemic lupus erythematosus.
They identified 22 immune cell populations and discovered previously unrecognized immune cell types in the kidneys of lupus patients. This study highlighted the importance of tissue-specific immune responses in autoimmune diseases, revealing how different immune cell subsets interact within the affected organ. The findings provided new insights into the pathogenesis of lupus nephritis and demonstrated the power of mass cytometry in unraveling complex tissue-resident immune landscapes in autoimmune conditions.
A pivotal study by Nehar-Belaid et al. (2020) in Nature Immunology, “Mapping systemic lupus erythematosus heterogeneity at the single-cell level,” used flow cytometry alongside other single-cell technologies to create a comprehensive atlas of immune cell states in SLE. They identified distinct subsets of effector CD4+ T cells and activated B cells associated with disease activity, providing new targets for therapeutic intervention.
Type 1 Diabetes: Biomarker Discovery
In type 1 diabetes (T1D), mass cytometry has been pivotal in identifying potential biomarkers for disease progression and treatment response.
Wang et al. (2019) published a significant paper in Cell Metabolism, titled “Single-cell mass cytometry analysis of the human endocrine pancreas.” This study used a 37-marker mass cytometry panel to characterize the cellular composition of human pancreatic islets, including in the context of type 1 diabetes (T1D). The researchers profiled over 100,000 single cells from pancreata of non-diabetic and T1D donors, revealing novel subpopulations of endocrine cells and immune cells. Notably, they identified a unique immune cell signature in T1D islets, characterized by an increase in cytotoxic T cells and a decrease in regulatory T cells. This work provided unprecedented insights into the cellular landscape of the human endocrine pancreas in health and disease, offering potential new targets for therapeutic intervention in T1D.
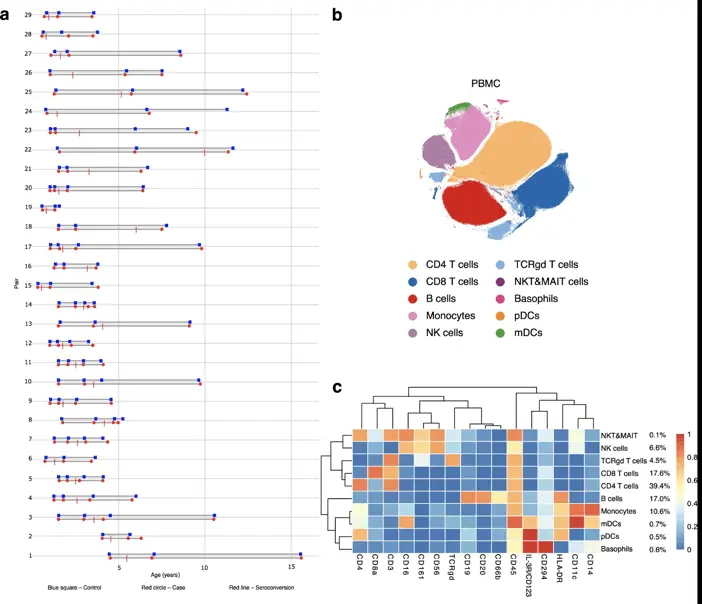
A study by Kallionpää et al. (May 2024) in Nature Communication, used mass cytometry to examine early immune responses in children who later develop type 1 diabetes. Analyzing blood samples from children categorized by their first-appearing autoantibodies, they found distinct immune cell compositions associated with different autoantibody profiles. Notably, they observed increased CD161 expression in natural killer cells of children with multiple autoantibodies. This research highlights the heterogeneity in type 1 diabetes progression and demonstrates mass cytometry’s potential in identifying biomarkers for disease development, potentially leading to more personalized prediction and prevention strategies.
Implications for Drug Discovery and Treatment
The insights gained from these mass cytometry studies have direct implications for drug discovery and treatment strategies:
- Targeting Tph cells: The identification of Tph cells in RA has led to new therapeutic strategies. While no drugs specifically targeting Tph cells have been approved yet, several are in preclinical development.
- Personalized treatment in SLE: The heterogeneity of immune cell states revealed in SLE suggests that personalized treatment approaches, tailored to an individual’s specific immune profile, may be more effective than current one-size-fits-all strategies.
- Immunomodulation in T1D: The characterization of regulatory T cells in T1D provides new targets for immunomodulatory therapies aimed at preserving beta cell function.
Mass cytometry has revolutionized our understanding of autoimmune diseases, revealing the complex cellular landscapes that underlie these conditions. From uncovering novel cell subsets in rheumatoid arthritis to mapping intricate cytokine networks in lupus and identifying potential biomarkers in type 1 diabetes, this technology has provided unprecedented insights into disease mechanisms.
As we continue to harness the power of mass cytometry, we move closer to a future where autoimmune diseases can be diagnosed earlier, monitored more accurately, and treated more effectively. The once opaque world of autoimmunity is gradually coming into focus, with mass cytometry serving as our high-resolution lens into this intricate biological realm. Each discovery brings us one step closer to turning the tide against these challenging diseases, offering hope to millions of patients worldwide.
When I dived into the world of autoimmune diseases with Esther, I realized immunology is like a vast ocean - every time you think you've seen it all, you discover a new depth. Our paper, "Proinflammatory T Helper 17 Cells Are Increased in Remission Rheumatoid Arthritis Patients and Triggered by Synergy Between Low-Dose Interleukin-7 and Interleukin-15" (Koppejan et al., 2022), was just the tip of the iceberg. Working on rheumatoid arthritis opened my eyes to the complexity of the immune system. But the real surprise? It became the perfect playground for developing Cytofast. Suddenly, I was collaborating with more people than I ever imagined possible. It was like hosting a scientific potluck where everyone brought their expertise to the table. Who knew that diving into autoimmune diseases would lead to creating a tool that brought the scientific community closer together? In the end, immunology didn't just teach me about cells - it taught me about connections, both cellular and human.
Guillaume Beyrend