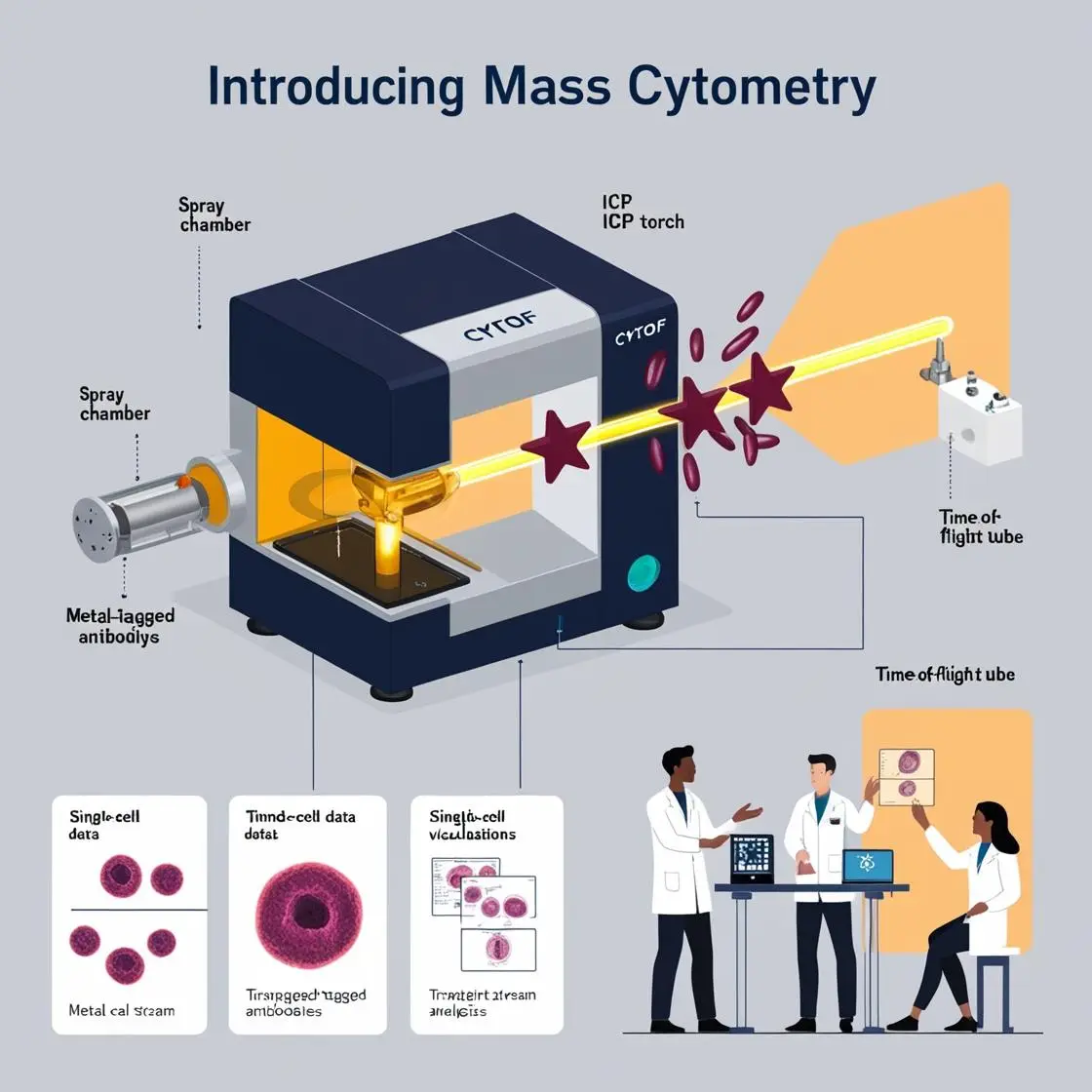
Imagine standing before a gleaming mass cytometer, its sleek exterior hiding a world of intricate technology within. This marvel of modern science, often affectionately called a “CyTOF” (Cytometry by Time of Flight), is the beating heart of mass cytometry. Let’s go ahead and take a journey through its components and workings. Would you like us to do it?
Components of a Mass Cytometer
At first glance, a mass cytometer might remind you of a particularly sophisticated coffee machine – but instead of brewing your morning espresso, it’s brewing groundbreaking science. Let’s break it down:
- Sample Introduction System: This is where our cellular adventure begins. Cells, carefully labeled with metal-tagged antibodies, are introduced into the system. It’s like a very tiny, complex race’s starting line.
- Nebulizer: Our cellular contestants are turned into a fine mist. Imagine a cellular spray tan booth.
- Plasma Torch: This is where things get hot – literally. The cell mist is vaporized and ionized in a plasma reaching temperatures of about 7000°C. That’s hotter than the surface of the sun! Our cells go from mist to plasma faster than you can say “cytokine storm.”
- Ion Optics: These lenses focus our newly formed ions, much like a microscope focuses light. It’s crowd control for ions, ensuring they behave and move in the right direction.
- Time-of-Flight Chamber: The heart of our CyTOF, where the magic happens. But more on this in a moment.
6. Detector: The finish line of our atomic race, where ions are counted and quantified.
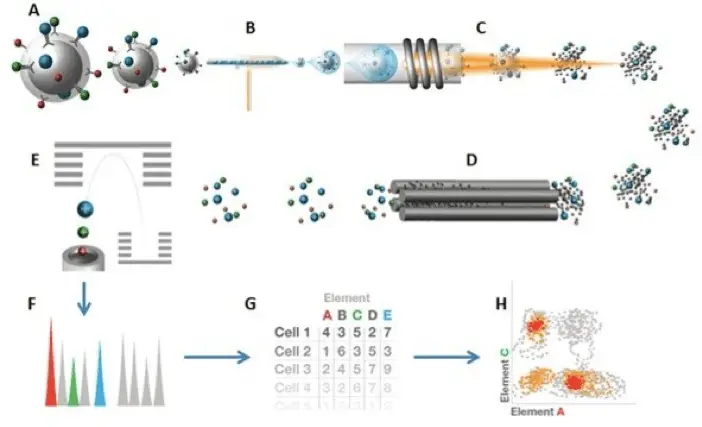
Time-of-Flight Mass Spectrometry
Now, let’s zoom in on that Time-of-Flight (TOF) chamber. This is where mass cytometry earns its name and its stripes.
Picture this: You’re at a track meet. Runners of different weights are lined up. The starting gun fires, and they’re off! Naturally, the lighter runners will reach the finish line first, followed by the heavier ones. This happens in the TOF chamber but with ions instead of runners.
The ions, created from our vaporized cells, are given a push with the same kinetic energy. They then fly through the chamber. Lighter ions (from more petite metal tags) reach the detector first, while heavier ones lag. By measuring each ion’s time to get the detector, we can determine its mass and, thus, which antibody it was attached to.
It’s like an atomic-scale Olympic sprint, where every millisecond counts!
Comparison of Different Mass Cytometry Platforms
While the basic principles remain the same, mass cytometry platforms have evolved, each with quirks and capabilities.
- The Original CyTOF is the firstborn of the mass cytometry world. When it debuted, it was like the first iPhone in its field—revolutionary but with room for improvement.
- CyTOF 2: These were the teenage years of mass cytometry. It was faster, more sensitive, and with better resolution—like upgrading from a flip phone to a smartphone.
- Helios is the current star of the show. With improved sensitivity and faster acquisition rates, it’s the Tesla of mass cytometers. One researcher described working with Helios as “like going from watching standard TV to 4K Ultra HD.”
- CyTOF XT is the new kid on the block. It boasts even faster acquisition rates and is more user-friendly. One scientist joked that it’s so easy to use that “even a principal investigator could run it!”. I haven’t had the chance to run any sample on it though, so I cannot confirm…

Anecdotes from the Field
I once heard a colleague’s story about their first time using a CyTOF. They had carefully prepared their samples, designed their panel, and were ready. They pressed start, and… nothing happened. Panic set in. After a frantic call to tech support, they realized they had forgotten to turn on the argon gas—the equivalent of trying to drive a car without gas! It’s a reminder that even the most sophisticated machines still need the basics.
Speaking of basics, I have my embarrassing tale to share. On my first day with our new CyTOF, I spent an agonizing 10 minutes searching for the computer’s “ON” button. It turns out high-tech doesn’t always mean intuitive! Another time, I was doing the maintenance of the CyTOF and only a few people were trained to do so. Everything was perfect, but I couldn’t get the machine running. I had simply forgotten a tissue inside the machine. The engineer who came from Germany to solve our problem was not disappointed by his trip from Germany to Leiden.
Mass Cytometry vs. Flow Cytometry: A Comparative Look
While mass cytometry has revolutionized high-dimensional single-cell analysis, it’s essential to understand how it compares to its older sibling, flow cytometry. Let’s break down some key differences:
- Acquisition Time and Flow Rate:
– Flow Cytometry: Lightning fast. An average sample takes about 2 minutes to acquire, with flow rates typically ranging from 2,000 to 10,000 events per second.
Mass Cytometry is more of a marathon runner. Expect around 30 minutes per sample, with 300-500 events per second flow rated. It’s the tortoise to flow cytometry’s hare, but remember the moral of that fable!
- Sample Size:
– Flow Cytometry: Can handle larger sample sizes, often millions of cells.
– Mass Cytometry: Typically works with smaller samples, usually around 500,000 to 1 million cells.
- Number of Parameters:
– Flow Cytometry: Generally limited to 15-30 parameters due to spectral overlap.
– Mass Cytometry: Can measure 40+ parameters simultaneously with minimal signal overlap.
- Cell Viability:
– Flow Cytometry: Cells remain viable and can be sorted for further analysis.
– Mass Cytometry: Cells are vaporized during analysis. It’s a one-way ticket for your samples!
- Cost and Availability:
Flow Cytometry is more affordable and widely available. A typical flow cytometer might cost $200,000-$500,000.
Mass Cytometry requires a significant investment. A CyTOF system can cost upwards of $600,000-$800,000.
Regarding availability, flow cytometers are ubiquitous in research institutions worldwide. You’d need one to find a significant biology department. Mass cytometers, while growing in popularity, are still relatively rare. As of 2021, there were estimated to be around 200 CyTOF instruments in North America and about 100 in Europe. In contrast, there are tens of thousands of flow cytometers globally.
This disparity in availability means that while flow cytometry is a daily tool for many immunologists, getting time on a mass cytometer often requires careful planning and sometimes travel. Researchers are making pilgrimages across countries to run their samples on a CyTOF!
Despite these challenges, mass cytometry’s unique capabilities make it an invaluable tool for many researchers. One colleague said, “Flow cytometry is like painting with primary colors. Mass cytometry lets you use the entire palette.”
While mass cytometry may be slower and less widely available than flow cytometry, its ability to provide high-dimensional data at the single-cell level makes it an indispensable tool in the modern immunologist’s arsenal. Whether using flow or mass cytometry, remember that every cell tells a story; these machines are our high-tech storytellers.
Imagine a tiny room, a million-dollar machine humming away, and... the sweet strains of a violin? Welcome to my CyTOF world, where Beethoven meets biotech. Running samples on this beast is like babysitting a temperamental toddler - you can't look away for a second. Facing 8-10 days of 14-hour shifts in a glorified closet, I needed a sanity saver. Enter my trusty violin. The soundproof room turned out to be a miniature concert hall. There I was, lab coat on, bow in hand, looking like I was auditioning for "America's Got Talent: Mad Scientist Edition." My colleagues couldn't resist snapping a photo - for posterity, or perhaps blackmail. But those marathon sessions flew by. Between watching for clogs and tuning my A string, time became a mere concept. And hey, how many scientists can say they've serenaded their samples? So next time you see a neatly plotted graph, remember - behind that data might be a scientist with a violin, turning the scientific method into a symphony. Who says you can't culture cells and yourself at the same time?
Guillaume Beyrend